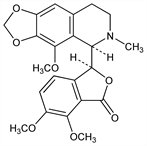
6718. Noscapine
Nomenclature
Description and references
An opium alkaloid, isolated from the plant Papaver somniferum L. Papaveraceae. Present in amounts up to 11% depending on season and locality. First isoln: Robiquet, Ann. Chim. Phys. [2] 5, 275 (1817). Extractable from the water-insoluble residue remaining from the processing of opium for the manufacture of morphine. Racemization to gnoscopine: Rabe, McMillian, Ann. 377, 233 (1910); and structural studies: Perkin, Robinson, J. Chem. Soc. 99, 775 (1911); Marshall et al., ibid. 1934, 1318. Preliminary stereochemical studies: Ohta et al., Tetrahedron Lett. 1963, 1857; Battersby, Spenser, J. Chem. Soc. 1965, 1087. Revised stereochemistry: Blaha et al., Collect. Czech. Chem. Commun. 29, 2328 (1964); Snatzke et al., Tetrahedron 25, 5059 (1969). Synthesis of racemate: Kerekes, Bognar, J. Prakt. Chem. 313, 923 (1971). Biosynthesis: Battersby, Hirst, Tetrahedron Lett. 1965, 669. Metabolism: N. Tsunoda, Y. Yoshimura, Xenobiotica 9, 181 (1979); eidem, ibid. 11, 23 (1981). Pharmacokinetics: B. Dhalstroem et al., Eur. J. Clin. Pharmacol. 22, 535 (1982). Clinical evaluation as antitussive: D. W. Empey et al., ibid. 16, 393 (1979). HPLC determn in serum: K. M. Jensen, J. Chromatogr. 274, 381 (1983). Comprehensive description: M. A. Al-Yahya, M. M. A. Hassan, Anal. Profiles Drug Subs. 11, 407-461 (1982).
Properties
Orthorhombic bisphenoidal prisms, tablets from diacetone. Triboluminescent. d 1.395. mp 176°. Sublimes at 150-160° under 11 mm pressure at 2 mm distance. Very weak base forming unstable salts with acids and strong bases. pK 7.8. uv max (ethanol): 209, 291, 309-310 nm (log ε 4.86, 3.60, 3.69). Practically insol in vegetable oils. Slightly sol in NH4OH, hot solns of KOH and NaOH, forming salts. Salts formed with acids are dextrorotatory and unstable in water.Derivative
Hydrochloride.Nomenclature
Properties
Hemihydrate to tetrahydrate, crystals, very sol in water forming basic salts.Derivative
Camphorsulfonate.Nomenclature
Properties
Contains 35.97% camphosulfonic acid. Prepn: Maillard, US 3108106 (1963 to Jacques Logeais). Crystals, mp 188-191°. [α]D33 +32.7° (c = 4.56 in water). Freely sol in water. Sol in methanol, ethanol. Slightly sol in ethyl acetate. Practically insol in ether.Derivative
dl-Form.Nomenclature
Properties
Long needles from methanol, mp 232° (dec). pK 7.8. Freely sol in carbon disulfide, hot chloroform; sol in ≈1500 parts alcohol; sparingly sol in benzene, water.Therapeutic Category
Antitussive.
Keywords
Antitussive