6731. Nybomycin
Nomenclature
CAS number: 30408-30-1
8-(Hydroxymethyl)-6,11-dimethyl-2H,4H-oxazolo[5,4,3-ij]pyrido[3,2-g]quinoline-4,10(11H)-dione; 6,11-dimethyl-8-(hydroxymethyl)pyrido[3,2-g]oxazolo[5,4,3-ij]quinoline-4,10(2H,11H)-dione. C16H14N2O4; mol wt 298.29.
C 64.42%, H 4.73%, N 9.39%, O 21.45%.
Description and references
Antibiotic substance produced by Streptomycete A 717 isolated from Missouri soil: Strelitz et al., Proc. Natl. Acad. Sci. USA 41, 620 (1955); Eble et al., Antibiot. Chemother. 8, 627 (1958); Brock, Sokolski, ibid. 631. Structure: Rinehart, Renfroe, J. Am. Chem. Soc. 83, 3729 (1961). Revised structure: Rinehart et al., ibid. 92, 6994 (1970). Total synthesis of deoxynybomycin: Forbis, Rinehart, ibid. 6995. Total synthesis of nybomycin: eidem, J. Antibiot. 24, 326 (1971); eidem, J. Am. Chem. Soc. 95, 5003 (1973).
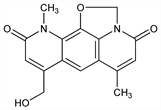
Properties
Needles from acetic acid, mp 325-330°. Sublimes at 250° (15 mm). Optically inactive. uv max (ethanol): 266, 285 nm. Soluble in concd acids. Very slightly sol in water, alkalies, and common organic solvents. Shows antiphage and antibacterial properties. LD50 i.p. in mice: 650 mg/kg (Brock, Sokolski). 13C NMR spectrum: A. M. Nadzan, K. L. Rinehart, J. Am. Chem. Soc. 99, 4647 (1977).Derivative
Acetate.C18H16N2O5; mol wt 340.33.
C 63.52%, H 4.74%, N 8.23%, O 23.51%.
Properties
Crystals from chloroform + ethanol, mp 236-237°.Derivative
Succinate.C20H19N2O7; mol wt 399.37.
C 60.15%, H 4.80%, N 7.01%, O 28.04%.