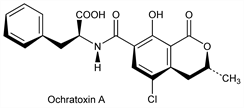
6740. Ochratoxins
Description and references
Toxic metabolites from Aspergillus ochraceus Wilh.: Scott, Mycopathol. Mycol. Appl. 25, 213 (1965); A. sulphureus and A. melleus: Lai et al., Appl. Microbiol. 19, 542 (1970); and Penicillium viridicatum Westling: van Walbeek et al., Can. J. Microbiol. 15, 1281 (1969). As these molds occur widely, some toxins have been found as natural contaminants on corn, peanuts, storage grains, cottonseed and other decaying vegetation. Isolns: van Walbeek et al., ibid. 14, 131 (1968); Shotwell et al., Appl. Microbiol. 17, 765 (1969); Nesheim, J. Assoc. Off. Anal. Chem. 52, 975 (1969); Natori et al., Chem. Pharm. Bull. 18, 2259 (1970). Structure and stereochemistry: van der Merwe et al., J. Chem. Soc. 1965, 7083; Nature 205, 1112 (1965). Synthesis of ochratoxins A, B and ester deriv: Steyn, Holzapfel, Tetrahedron 23, 4449 (1967); Nesheim, loc. cit.; Roberts, Woolven, J. Chem. Soc. C 1970, 278. Facile synthesis of A: G. A. Kraus, J. Org. Chem. 46, 201 (1981). Toxicity study: Purchase, Theron, Food Cosmet. Toxicol. 6, 479 (1968). Physicochemical data: A. E. Pohland et al., Pure Appl. Chem. 54, 2220 (1982). Comprehensive reviews: Steyn, “Ochratoxin and other Dihydroisocoumarins” in Microbial Toxins vol. VI, A. Ciegler, et al., Eds. (Academic Press, New York, 1971) p 179-205; F. S. Chu, Crit. Rev. Toxicol. 2, 499-524 (1974); P. Krogh in Natural Toxins, D. Eaker, P. Wadstr"om, Eds. (Pergamon, New York, 1980) pp 673-680; A. E. Pohland et al., Pure Appl. Chem. 64, 1029-1046 (1992).