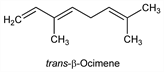
6741. Ocimene
Nomenclature
Description and references
From the leaves of Ocimum basilicum L., Labiatae; Baronia dentigeroides Cheel, Rutaceae; Litsea zeylanica C. & T. Nees, Lauraceae; Homoranthus flavescens A. Cunn., Myrtaceae. From the fruits of Evodia rutaecarpa (Juss.) Hook. f. & Thoms., Rutaceae. Exists in two modifications: α-form, 3,7-dimethyl-1,3,7-octatriene; and β-form, 3,7-dimethyl-1,3,6-octatriene. Cis and trans refers to the stereochemistry at the double bond between positions 3 and 4. Isoln and structure: van Romburgh, Proc. K. Ned. Akad. Wet. 3, 454 (1900); Enklaar, Rec. Trav. Chim. 26, 157 (1907); 27, 422 (1908); 36, 215 (1917); 45, 337 (1926). Structure: Sutherland, J. Am. Chem. Soc. 74, 2688 (1952). Separation of isomers: Ohloff et al., Ann. 675, 83 (1964). Synthesis of trans-α-form: O. P. Vig et al., Indian J. Chem. 7, 1111 (1969). Stereospecific synthesis: O. P. Vig et al., ibid. 15B, 25 (1977).