6827. Oleanolic Acid
Nomenclature
CAS number: 508-02-1
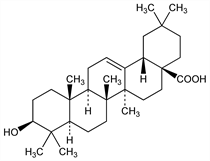
(3β)-3-Hydroxyolean-12-en-28-oic acid; oleanol; caryophyllin. C30H48O3; mol wt 456.70.
C 78.90%, H 10.59%, O 10.51%.
Description and references
Occurs in the free state in leaves of Olea europaea, Oleaceae, in leaves of Viscum album L., Loranthaceae, in buds of Syzygium aromaticum (L.) Merr. & Perry, Myrtaceae (cloves), in Swertia japonica (Maxim.) Makino, and in Centaurium umbellatum Gilib. (Erythraea centaurium (L.) Pers.), Gentianaceae; as acetate in birch bark, as glycoside in many saponins. Isoln procedures (from cloves): Winterstein, Stein, Z. Physiol. Chem. 202, 222 (1931); Ruzicka, Hofmann, Helv. Chim. Acta 19, 114 (1936); Picard et al., J. Chem. Soc. 1939, 1047. Structure: Ruzicka et al., Helv. Chim. Acta 29, 210 (1946). Review: J. Simonsen, W. C. T. Ross, The Terpenes vol. 5 (University Press, Cambridge, 1957) pp 221-285. Cf. α- and β-amyrin.
Properties
Fine, solvated needles from alc. After drying, mp 310°. [α]D20 +83.3° (c = 0.6 in chloroform). pK 2.52. Insol in water. Sol in 65 parts ether, 106 parts 95% alcohol, 35 parts boiling 95% alcohol, 118 parts chloroform, 180 parts acetone, 235 parts methanol.Derivative
Acetate.C32H50O4; mol wt 498.74.
C 77.06%, H 10.10%, O 12.83%.
Properties
Needles from methanol, mp 268°. [α]D17 +74.5° (c = 0.6 in CHCl3).Derivative
Methyl ester.C31H50O3; mol wt 470.73.
C 79.10%, H 10.71%, O 10.20%.
Properties
mp 201°. [α]D20 +75° (c = 0.6 in CHCl3).Derivative
Acetate of methyl ester.C33H52O4; mol wt 512.76.
C 77.30%, H 10.22%, O 12.48%.