6874. Ornithine
Nomenclature
Description and references
Non-essential amino acid for human development but is required intermediate in arginine biosynthesis. Found in virtually all vertebrate tissues as well as incorporated into proteins, such as tyrocidine, q.v. Isolation from chicken excreta: M. Jaffe, Ber. 10, 1925 (1877). Early chemistry and biochemistry: J. P. Greenstein, M. Winitz, Chemistry of the Amino Acids (John Wiley and Sons, Inc., New York, 1961) pp. 2477-2491, passim. Review of synthesis: R. Yoshida, Synth. Prod. Util. Amino Acids, T. Kaneko et al., Eds (Kodansha Ltd, Tokyo, Japan, 1974) pp 166-170; of biosynthesis in plants: P. D. Shargool et al., Phytochemistry 27, 1571-1574 (1988). Brief historical review: H. A. Akers, E. V. Dromgoole, Trends Biochem. Sci. 7, 156-157 (1982). Review of metabolism: V. E. Shih, Enzyme 26, 254-258 (1981); of role in CNS functions: N. Seiler, G. Daune-Anglard, Metab. Brain Dis. 8, 151-179 (1993).
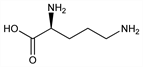
Properties
Colorless crystals. [α]D25 +11.5° (c = 6.5). Aq solns are alkaline. pK1′ 1.94; pK2′ 8.65; pK3′ 10.76. Freely sol in water, alc; sparingly sol in ether.Derivative
l-Form aspartate.Nomenclature
Properties
Crystal structure: D. M. Salunke, M. Vijayan, Int. J. Pept. Protein Res. 22, 154 (1983). Clinical trial in hyperammonemia: J. Fehér et al., Med. Sci. Monit. 3, 669 (1997); S. Stauch et al., J. Hepatol. 28, 856 (1998).Derivative
dl-Form.Nomenclature
Properties
Crystals from water. Sparingly sol in alcohol.Therapeutic Category
In treatment of hyperammonemia.