6883. Orthocaine
Nomenclature
CAS number: 536-25-4
3-Amino-4-hydroxybenzoic acid methyl ester; methyl 3-amino-4-hydroxybenzoate; Orthoform (Winthrop); Orthoform New. C8H9NO3; mol wt 167.16.
C 57.48%, H 5.43%, N 8.38%, O 28.71%.
Description and references
Prepd by dissolving 3-amino-4-hydroxybenzoic acid in methanol, saturating with HCl gas: Einhorn, Pfyl, Ann. 311, 46 (1900); DE 97333; Chem. Zentralbl. 1898, II, 525. Also prepd by reduction of methyl 3-nitro-4-hydroxybenzoate with aluminum amalgam: Auwers, R"ohrig, Ber. 30, 991 (1897); with tin and HCl or with stannous chloride and alcoholic HCl: DE 97334; Chem. Zentralbl. 1898, II, 526.
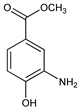
Properties
Needles from benzene, mp 143°. When crystallized from chloroform it sometimes assumes an allotropic form, mp 111°, which on melting changes to the normal form and after solidifying always melts at 143°. Odorless and tasteless. Neutral reaction. Almost insol in cold water. Moderately sol in hot water with gradual decompn forming 3-amino-4-hydroxybenzoic acid and methanol. One gram dissolves in 6 ml alcohol, 50 ml ether; readily dissolves in aq NaOH. Forms water sol salts with HCl and HBr.Note
The name Orthoform was applied originally to the methyl ester of 4-amino-3-hydroxybenzoic acid which was also used as a local anesthetic. This compd now is designated as Orthoform Old.Therapeutic Category
Anesthetic (topical).
Therapeutic Category (Veterinary)
Anesthetic (topical).Keywords
Anesthetic (Local)