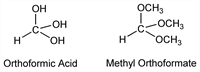
6884. Orthoformic Acid
Nomenclature
CAS number: 463-78-5
Methanetriol; trihydroxymethane. CH4O3; mol wt 64.04.
C 18.75%, H 6.30%, O 74.95%.
Description and references
Hypothetical compd, illustrated for nomenclature purposes only. The hydrogens of the hydroxyl groups are replaceable with alkyl groups giving rise to orthoformic esters, which are real compds (produced by various syntheses, e.g. from sodium alcoholates and chloroform). Monograph: H. W. Post, The Chemistry of the Aliphatic Orthoesters (Reinhold, New York, 1943), 188 pp.
Derivative
Trimethyl ester.Nomenclature
CAS number: 149-73-5
Trimethoxymethane; methyl orthoformate. C4H10O3; mol wt 106.12.
C 45.27%, H 9.50%, O 45.23%.
Description and references
Prepd from chloroform and methanol in presence of sodium: Sah, Ma, J. Am. Chem. Soc. 54, 2965 (1932).
Properties
Liquid. d420 0.9676; d425 0.9623. bp760 100.6°. nD25 1.3773.Derivative
Triethyl ester.Nomenclature
CAS number: 122-51-0
1,1′,1″-[Methylidynetris(oxy)]tris[ethane]; ethyl orthoformate; triethoxymethane; triethyl orthoformate; Aethone (Sopar). C7H16O3; mol wt 148.20.
C 56.73%, H 10.88%, O 32.39%.
Description and references
Prepd from chloroform and ethanol in presence of sodium: Chu, Shen, C.A. 38, 29307 (1944). Toxicity study: H. F. Smyth et al., Arch. Ind. Hyg. Occup. Med. 4, 119 (1951).