6933. Oxethazaine
Nomenclature
CAS number: 126-27-2
2,2′-[(2-Hydroxyethyl)imino]bis[N-(1,1-dimethyl-2-phenylethyl)-N-methylacetamide]; N,N-bis[N-methyl-N-phenyl-tert-butylacetamido]-β-hydroxyethylamine; oxetacaine; oxethazine; Wy-806; Storocain (Eisai); Topicain (Chugai). C28H41N3O3; mol wt 467.64.
C 71.91%, H 8.84%, N 8.99%, O 10.26%.
Description and references
Prepn: Seifter et al., US 2780646 (1957 to Am. Home Prods.); Salomon, IL 10062 (1957), C.A. 52, 15569 (1958). Pharmacology: Axerio Agnessetti, An. R. Acad. Farm. 35, 183 (1969), C.A. 71, 122082q (1969). Toxicity studies: J. M. Glassman et al., Toxicol. Appl. Pharmacol. 5, 184 (1963). Clinical trial of combination with antacid in ulcer pain: F. T. M. Cordeiro et al., Curr. Ther. Res. 19, 230 (1976); A. Zanni et al., Curr. Med. Res. Opin. 10, 128 (1986). Evaluation as dental anesthetic: P. A. Brennan, J. D. Langdon, Br. J. Oral Maxillofac. Surg. 28, 26 (1990).
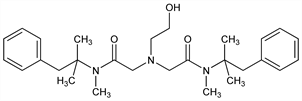
Properties
Crystals from benzene + hexane, mp 104-104.5°. Insol in water. Sol in dil acids.Derivative
Hydrochloride.Nomenclature
CAS number: 13930-31-9
Emoren (Wassermann). C28H41N3O3.HCl; mol wt 504.10.
C 66.71%, H 8.40%, N 8.34%, O 9.52%, Cl 7.03%.
Properties
mp 146-147°. Easily sol in water. LD50 (calculated as base) in mice, rats (mg/kg): 399.9, 625.9 orally; 247.2, 502.3 i.m.; 3.6, 1.3 i.v.; LD50 (calculated as base) in rabbits (mg/kg): 0.54 i.v. (Glassman).Derivative
Combination with aluminum and magnesium hydroxides.Nomenclature
Mutesa (Wyeth); Muthesa (Wyeth); Oxaine M (Wyeth); Tepilta (Wyeth).Therapeutic Category
Anesthetic (local).
Keywords
Anesthetic (Local)