6945. Oxolinic Acid
Nomenclature
CAS number: 14698-29-4
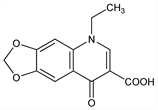
5-Ethyl-5,8-dihydro-8-oxo-1,3-dioxolo[4,5-g]quinoline-7-carboxylic
acid; 1-ethyl-1,4-dihydro-6,7-methylenedioxy-4-oxo-3-quinolinecarboxylic
acid; 1-ethyl-6,7-methylenedioxy-4-quinolone-3-carboxylic acid; W-4565; Inoxyl (Debat); Nidantin (Warner-Lambert); Uritrate (Warner-Lambert); Urotrate (Warner-Lambert). C13H11NO5; mol wt 261.23.
C 59.77%, H 4.24%, N 5.36%, O 30.62%.
Description and references
Quinolone antibacterial. Prepn: Kaminsky, Meltzer, US 3287458 (1966 to Warner-Lambert); eidem, J. Med. Chem. 11, 160 (1968). Pharmacology: Turner et al., Antimicrob. Agents Chemother. 1967, 475 sqq. Metabolism studies: DiCarlo et al., Arch. Biochem. Biophys. 127, 503 (1968); Crew et al., Xenobiotica 1, 193 (1971). Use in urinary tract infections: S. Kalowski et al., Med. J. Aust. 21, 345 (1979); E. Anza et al., Minerva Med. 70, 2333 (1979). Mechanism of action study: H. T. Wright et al., Science 213, 455 (1981). Brief review of therapeutic use: R. Glickman et al., Am. J. Hosp. Pharm. 36, 1077-1079 (1979).
Properties
Crystals from DMF, mp 314-316° (dec). LD50 in mice, rats (mg/kg): >6000, >2000 orally (Turner).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Synthetic); Quinolones and Analogs