6947. Oxonic Acid
Nomenclature
CAS number: 937-13-3
1,4,5,6-Tetrahydro-4,6-dioxo-1,3,5-triazine-2-carboxylic acid; allantoxanic acid; 5-azaorotic acid; s-triazine-2,4-dione-6-carboxylic acid; 2,4-dioxo-1,2,3,4-tetrahydro-1,3,5-triazine-6-carboxylic acid. C4H3N3O4; mol wt 157.08.
C 30.58%, H 1.93%, N 26.75%, O 40.74%.
Description and references
Formed during alkaline oxidation of uric acid with H2O2 or KMnO4. Prepn and structure: Brandenberger, Helv. Chim. Acta 37, 641 (1954); Brandenberger, Brandenberger, ibid. 37, 2207 (1954); Piskala, Gut, Collect. Czech. Chem. Commun. 27, 1572 (1962). Metabolism: Chelbova et al., Biochem. Pharmacol. 19, 2785 (1970); Cihak, Sorm, ibid. 21, 607 (1972).
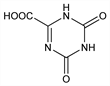
Properties
Free acid is very unstable; can be isolated as the monopotassium salt. uv max (pH 9): 255 nm (ε 6800); (pH 12): 252 nm (ε 4600).Derivative
Oxonic acid amide.C4H4N4O3; mol wt 156.10.
C 30.78%, H 2.58%, N 35.89%, O 30.75%.