6977. Oxythiamine
Nomenclature
CAS number: 582-36-5
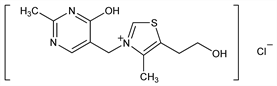
3-[(1,4-Dihydro-2-methyl-4-oxo-5-pyrimidinyl)methyl]-5-(2-hydroxyethyl)-4-methylthiazolium
chloride; 5-(2-hydroxyethyl)-3-[(4-hydroxy-2-methyl-5-pyrimidinyl)methyl]-4-methylthiazolium
chloride. C12H16ClN3O2S; mol wt 301.79.
C 47.76%, H 5.34%, Cl 11.75%, N 13.92%, O 10.60%, S 10.62%.
Description and references
Prepn: F. Bergel, A. R. Todd, J. Chem. Soc. 1937, 1504; M. Soodak, L. R. Cerecedo, J. Am. Chem. Soc. 66, 1988 (1944). Improved prepn: H. N. Rydon, Biochem. J. 48, 383 (1951). Thiamine antagonist activity: A. J. Eusebi, L. R. Cerecedo, Science 110, 162 (1949); L. J. Daniel, L. C. Norris, Proc. Soc. Exp. Biol. Med. 72, 165 (1949); and distribution in tissues: C. J. Gubler, D. S. Murdock, J. Nutr. Sci. Vitaminol. 28, 217 (1982). Proposed mechanism of action: S. A. Strumilo et al., Biomed. Biochim. Acta 43, 159 (1984). Determn by HPLC: B. C. Hemming, C. J. Gubler, J. Liq. Chromatogr. 3, 1697 (1980).
Derivative
Hydrochloride.C12H16ClN3O2S.HCl; mol wt 338.25.
C 42.61%, H 5.07%, Cl 20.96%, N 12.42%, O 9.46%, S 9.48%.