7012. Pantethine
Nomenclature
CAS number: 16816-67-4
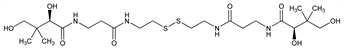
(2R,2′R)-N,N′-[Dithiobis[2,1-ethanediylimino(3-oxo-3,1-propanediyl)]]bis[2,4-dihydroxy-3,3-dimethylbutanamide]; N,N′-[dithiobis(ethyleneiminocarbonylethylene)]bis(2,4-dihydroxy-3,3-dimethylbutyramide); d-bis(N-pantothenyl-β-aminoethyl) disulfide; Lipodel (Delalande); Pantetina (Maggioni); Panthecin (Sawai); Pantomin (Daiichi); Pantosin (Daiichi). C22H42N4O8S2; mol wt 554.72.
C 47.63%, H 7.63%, N 10.10%, O 23.07%, S 11.56%.
Description and references
Disulfide dimer of pantetheine, q.v. Growth factor for Lactobacillus bulgaricus: Williams et al., J. Biol. Chem. 177, 933 (1949) PubMed. Formed by oxidation of pantetheine: Brown, Snell, ibid. 198, 375 (1952) PubMed. Structure: Snell et al., J. Am. Chem. Soc. 72, 5349 (1950) DOI. Synthesis: Wieland, Bokelmann, Naturwissenschaften 38, 384 (1951) DOI; Wittle et al., J. Am. Chem. Soc. 75, 1694 (1953) DOI; Viscontini et al., Helv. Chim. Acta 37, 375 (1954) DOI; Bowman, Cavalla, J. Chem. Soc. 1954, 1171 DOI; Shimizu et al., Chem. Pharm. Bull. 13, 180 (1965) PubMed. Clinical trial in hyperlipoproteinemia: A. Gaddi et al., Atherosclerosis 50, 73 (1984) DOI PubMed.
Properties
Glassy, colorless to light yellow substance. [α]D27 +13.5° (c = 3.75 in water). Freely sol in water; less sol in ethanol. Practically insol in ether, acetone, ethyl acetate, benzene, chloroform.Therapeutic Category
Antilipemic.
Keywords
Antilipemic