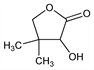
7013. Pantolactone
Nomenclature
CAS number: 599-04-2
Dihydro-3-hydroxy-4H-dimethyl-2(3H)-furanone; pantoic acid γ-lactone; pantoyl lactone; pantoic lactone; 2,4-dihydroxy-3,3-dimethylbutyric acid γ-lactone; α-hydroxy-β,β-dimethyl-γ-butyrolactone. C6H10O3; mol wt 130.14.
C 55.37%, H 7.75%, O 36.88%.
Description and references
A degradation product of pantothenic acid from liver: Williams, Major, Science 91, 246 (1940). Important intermediate in the synthesis of pantothenic acid. May be prepd by condensing isobutyraldehyde with formaldehyde yielding α,α-dimethyl-β-hydroxypropionaldehyde which is condensed with hydrocyanic acid in the presence of calcium chloride to form racemic pantolactone. Various modifications of this procedure exist: Glaser, Monatsh. Chem. 25, 46 (1904); Stiller et al., J. Am. Chem. Soc. 62, 1785 (1940); Reichstein, Grüssner, Helv. Chim. Acta 23, 650 (1940); Carter, Ney, J. Am. Chem. Soc. 63, 312 (1941). Vast patent literature, e.g., Beckmann et al.; Klein, US 2967869 and US 3024250 (1961, 1962, both to Nopco).