7017. Papaveraldine
Nomenclature
CAS number: 522-57-6
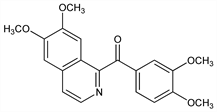
(6,7-Dimethoxy-1-isoquinolinyl)(3,4-dimethoxyphenyl)methanone; 6,7-dimethoxy-1-isoquinolyl 3,4-dimethoxyphenyl ketone; 6,7-dimethoxy-1-veratroylisoquinoline; xanthaline. C20H19NO5; mol wt 353.37.
C 67.98%, H 5.42%, N 3.96%, O 22.64%.
Description and references
The old name papaveraldine is retained to avoid confusion. From opium; whether papaveraldine occurs as such in the poppy plant, or is formed during the process of extraction, has not been investigated. Oxidation of papaverine to papaveraldine by SeO2: Menon, Proc. Indian Acad. Sci. 19A, 21 (1944). For older references see: Small, Lutz, “Chemistry of the Opium Alkaloids,” in U.S. Public Health Reports Suppl. No. 103 (Washington, 1932). Synthesis from Reissert compds: Popp, McEwen, J. Am. Chem. Soc. 79, 3773 (1957).
Properties
Crystals from abs ethanol, mp 208-209°. Sol in benzene, chloroform; slightly sol in alcohol, ether, petr ether; nearly insol in water, alkalies or carbonates.Derivative
Hydrochloride.C20H19NO5.HCl; mol wt 389.83.
C 61.62%, H 5.17%, N 3.59%, O 20.52%, Cl 9.09%.