7088. Penicillamine
Nomenclature
Description and references
Characteristic degradation product of penicillin type antibiotics. Active as copper chelating agent and as a disease modifying antirheumatic drug (DMARD). Prepn by hydrolysis of penicillins: E. P. Abraham et al., Nature 151, 107 (1943). Review of syntheses of dl-form and enantiomers: H. M. Crooks in The Chemistry of Penicillin, H. T. Clarke et al., Eds. (Princeton Univ. Press, 1949) pp 455-472; W. M. Weigert et al., Angew. Chem. Int. Ed. 14, 330 (1975). Polymorphism of d-form: J. A. G. Vidler, J. Pharm. Pharmacol. 28, 662 (1976). Toxicity data: Veis et al., Antibiotiki 14, 837 (1969). CE determn in pharmaceuticals: R. Gotti et al., J. Chromatogr. A 844, 361 (1999). General reviews: I. A. Jaffe in Pharmacological and Biochemical Properties of Drug Substances Vol. 2, M. E. Goldberg, Ed. (Am. Pharm. Assoc., Washington, DC, 1979) pp 465-478; C. C. Chiu, L. T. Grady, Anal. Profiles Drug Subs. 10, 601-637 (1981). Review of assay methods: N. Kucharczyk, S. Shahiniam, J. Rheumatol. 8, Suppl. 7, 28-34 (1981); of metabolism and pharmacology: D. Perrett, ibid. 41-50; of clinical pharmacokinetics: D. A. Joyce, Pharmacol. Ther. 42, 405-427 (1989). Clinical trial in Wilson's disease: A. Czlonkowska et al., J. Neurol. 243, 269 (1996). Review of clinical experience in rheumatoid arthritis: R. Munro, H. A. Capell, Br. J. Rheumatol. 36, 104-109 (1997).
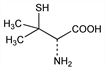
Properties
White or practically white, crystalline powder. mp 202-206° (Weigert). [α]D25 -63° (c = 0.1 in pyridine). Freely sol in water; slightly sol in alcohol. Insol in ether, acetone, benzene, carbon tetrachloride. LD50 in rats (mg/kg): >10000 orally, >660 i.p. (Jaffe).Derivative
Hydrochloride.Nomenclature
Properties
Hygroscopic crystals, dec 177.5°. [α]D25 -63° (1N NaOH). Freely sol in water, sol in ethanol. Aq solns are comparatively stable at pH 2-4. LD50 i.v. in mice: 2289 mg/kg (Veis).Derivative
dl-Form.Nomenclature
Properties
Crystals, dec 201°. pK: 1.8 (carboxyl); 7.9 (α-amino); 10.5 (β-thiol). LD50 orally in rats: 365 mg/kg (Jaffe).Derivative
dl-Form hydrochloride.Nomenclature
Properties
Crystals, dec 145-148°.Derivative
l-Form.Nomenclature
Properties
Crystals, mp 190-194°. [α]D25 +63° (in 1N NaOH). LD50 i.p. in rats: 350 mg/kg (Jaffe).Therapeutic Category
Antirheumatic. Chelating agent (copper); Wilson's Disease treatment.
Therapeutic Category (Veterinary)
Chelating agent to control hepatic copper levels in dogs.Keywords
Antiarthritic/Antirheumatic; Chelating Agent; Wilson's Disease Treatment