7094. Penicillin G
Nomenclature
Description and references
Discovery of antibiotic substance produced by Penicillium sp: A. Fleming, Br. J. Exp. Pathol. 10, 226 (1929). Preliminary isoln: P. W. Clutterbuck et al., Biochem. J. 26, 1907 (1932); and chemotherapeutic properties: E. Chain et al., Lancet 2, 226 (1940); E. P. Abraham et al., ibid. 2, 177 (1941). Review of early studies: E. Chain, Annu. Rev. Biochem. 17, 657-704 (1948); H. T. Clarke et al., The Chemistry of Penicillin (Princeton Univ. Press, 1949) 1094 pp. Crystal structure: D. C. Hodgkin, Adv. Sci. 6, 85 (1949). Fermentation process: A. L. Demain, N. L. Somerson, US 3024169 (1962 to Merck & Co.). Total synthesis: J. C. Sheehan, K. R. Henery-Logan, J. Am. Chem. Soc. 81, 5838 (1959); R. A. Firestone et al., J. Org. Chem. 39, 437 (1974). Review of clinical pharmacokinetics of penicillins: M. Barza, L. Weinstein, Clin. Pharmacokinet. 1, 297 (1976). Comprehensive description of the potassium salt: J. Kirschbaum, Anal. Profiles Drug Subs. 15, 427-507 (1987).
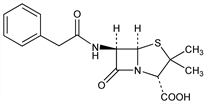
Properties
Amorphous white powder. [α]D +282° (ethanol). Sparingly sol in water. Sol in methanol, ethanol, ether, ethyl acetate, benzene, chloroform, acetone. Insol in petr ether.Derivative
Sodium salt.Nomenclature
Properties
Crystals from methanol + ethyl acetate. [α]D24.8 +301° (c = 2.0 in water). uv max (water): 252, 257.5, 264 nm (EM about 300, 240, 180). Freely sol in water, isotonic saline, glucose solns. Sol in methanol; less sol in ethanol. Practically insol in acetone, chloroform, ether, ethyl acetate, fixed oils, liquid paraffin.Derivative
Potassium salt.Nomenclature
Properties
Crystals from aq butanol, mp 214-217° (dec). Moderately hygroscopic. [α]D22 +285° (c = 0.748 in water). Freely sol in water, isotonic saline, glucose solns; sparingly sol in ethanol. Practically insol in chloroform, ether, fixed oils, liquid paraffin. pH of 6% aq soln 5.0 to 7.5.Derivative
Mixture with clemizole.Nomenclature
Description and references
Repository form of penicillin. Prepn: H. Mückter et al., Arzneim.-Forsch. 4, 487 (1954).
Properties
White powder, mp 144-145°. [α]D24 +144.5° (c = 10 in DMF). Sol in methanol, ethanol, DMF; slightly sol in water, acetone, dioxane.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins