7098. Penicillin N
Nomenclature
CAS number: 525-94-0
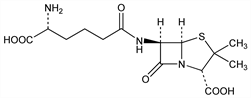
(2S,5R,6R)-6-[[(5R)-5-Amino-5-carboxy-1-oxopentyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; 6-(d-5-amino-5-carboxyvaleramido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; (d-4-amino-4-carboxybutyl)penicillinic acid; cephalosporin N; adicillin; Synnematin B. C14H21N3O6S; mol wt 359.40.
C 46.79%, H 5.89%, N 11.69%, O 26.71%, S 8.92%.
Description and references
Antibiotic substance produced by Cephalosporium spp found in sewage outpours: Gottshall et al., Proc. Soc. Exp. Biol. Med. 76, 307 (1951); Abraham et al., Nature 171, 343 (1953); 176, 551 (1955). Produced also by Paecilomyces persicimus: Pisano et al., Antimicrob. Agents Annu. 1960 (Plenum Press, New York, 1961) pp 41, 48; by Penicillium chrysogenum: Flynn et al., J. Am. Chem. Soc. 84, 4594 (1962). Structure: Abraham, Newton, Biochem. J. 58, 103 (1954). Production: Miller et al., US 2831797 (1958). Purification: Goodall, Sutcliffe, US 2899425 (1959 to ICI).
Properties
Soluble in water. Dextrorotatory. Inactivated by penicillinase as is penicillin G, but differs from the common peni-cillin by its antibacterial activity and hydrophilic character. When an aq soln is kept at pH 2.7 and 37° for 2 hrs, there is a loss of antibacterial activity and an increase in dextrorotation. Active against Sarcina lutea, Proteus vulgaris, Salmonella typhimurium, Diplococcus pneumoniae. Shows practically no activity against B. subtilis and Staph. aureus. The toxicity is somewhat less than that of penicillin G, although penicillin N is excreted more slowly.Derivative
Barium salt.Properties
White powder. [α]D20 +187° (c = 0.6). Freely sol in water, sparingly sol in methanol. Practically insol in ethanol.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins