7099. Penicillin O
Nomenclature
CAS number: 87-09-2
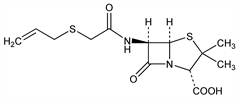
(2S,5R,6R)-3,3-Dimethyl-7-oxo-6-[[(2-propenylthio)acetyl]amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; [(allylthio)methyl] penicillin; allylmercaptomethylpenicillin; allylmercaptomethylpenicillinic acid; penicillin AT. C13H18N2O4S2; mol wt 330.42.
C 47.25%, H 5.49%, N 8.48%, O 19.37%, S 19.41%.
Description and references
Antibiotic produced by Penicillium chrysogenum. Biosynthesis of salts: Behrens et al., J. Biol. Chem. 175, 793 (1948); Rhodehamel, Behrens et al., US 2528175 and US 2623876 (1950, 1952, both to Lilly); Ford et al., Antibiot. Chemother. 3, 1149 (1953); Ford, US 2647894 (1953 to Upjohn); Palecková, Slechta, C.A. 50, 17309g (1956).
Derivative
2-Chloroprocaine salt monohydrate.Nomenclature
Chloroprocaine penicillin O; penicillin O 2-chloroprocaine; Depo-Cer-O-Cillin Chloroprocaine (Upjohn).C26H37ClN4O6S2.H2O; mol wt 619.19.
C 50.43%, H 6.35%, Cl 5.73%, N 9.05%, O 18.09%, S 10.36%.
Properties
Slender needles from hot water, mp 79-81°. Practically insol in cold water. Stable in dry form at room temp. Aq suspensions are stable at room temp for 1 week, at refrigerator temps for 3 weeks. Calculated activity: 949 units/mg. Solubilities: Weiss et al., Antibiot. Chemother. 7, 374 (1957).Derivative
Potassium salt.Nomenclature
CAS number: 897-61-0
Potassium penicillin O; penicillin O potassium. C13H17KN2O4S2; mol wt 368.51.
C 42.37%, H 4.65%, K 10.61%, N 7.60%, O 17.37%, S 17.40%.
Properties
Crystals from acetone. Soluble in water. Stable in dry form at room temp for at least 3 years. Requires no refrigeration when dry. Aq solns may be kept for 3 days at +10° without significant loss of activity. Behrens' prepn assayed 1630 units/mg. Less toxic than benzylpenicillin in exptl animals.Derivative
Procaine salt.C26H38N4O6S2; mol wt 566.73.
C 55.10%, H 6.76%, N 9.89%, O 16.94%, S 11.32%.
Properties
Crystals from water.Derivative
Sodium salt.Nomenclature
CAS number: 7177-54-0
Cer-O-Cillin Sodium (Upjohn). C13H17N2NaO4S2; mol wt 352.40.
C 44.31%, H 4.86%, N 7.95%, Na 6.52%, O 18.16%, S 18.20%.
Properties
Crystals from acetone.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins