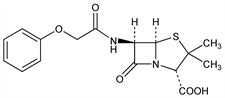
7100. Penicillin V
Nomenclature
Description and references
Obtained by adding 2-phenoxyethanol to the Penicillium culture using yeast autolyzate as source of nitrogen: Brandl et al., Wien. Med. Wochenschr. 1953, 602; Brandl, Margreiter, Oesterr. Chem.-Ztg. 55, 11-21 (1954), C.A. 48, 10296 (1954). Purification: Parker et al., J. Pharm. Pharmacol. 7, 683 (1953). Total synthesis: Sheehan, Henery-Logan, J. Am. Chem. Soc. 79, 1262 (1957); 81, 3089 (1959); 84, 2983 (1962). Prepn from 6-aminopenicillanic acid: Glambitza, Ann. 673, 166 (1964). Soly data: Weiss et al., Antibiot. Chemother. 7, 374 (1957). The biologically active form is the dextrorotatory d-form; the dl-form is half as active. l-Penicillin V has little, if any, antibiotic activity. Toxicity data: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Comprehensive description of the potassium salt: J. M. Dunham in Anal. Profiles Drug Subs. 1, 249-300 (1972); D. H. Sieh, ibid. 17, 677-748 (1988).
Properties
Crystals, dec 120-128°. Stable in air up to 37°; relatively stable to acid. uv max: 268, 274 nm (ε 1330, 1100). Soly in water at pH 1.8 (acidified with HCl) = 25 mg/100 ml. Sol in polar organic solvents. Practically insol in vegetable oils and in liquid petrolatum.Derivative
Potassium salt.Nomenclature
Properties
Soluble in water. [α]D25 +223° (c = 0.2). LD50 orally in rats: >1040 mg/kg (Goldenthal).Derivative
Calcium salt.Nomenclature
Derivative
Compd with dibenzylethylenediamine.Nomenclature
Description and references
Prepn: R. Brunner et al., US 2820789 (1958 to American Home Products).
Properties
Practically white powder having a characteristic odor, mp 105-109°. Soly at ≈28° (mg/ml): water 0.321; ethanol 14.6.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins