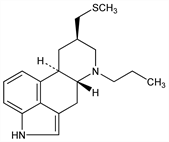
7162. Pergolide
Nomenclature
Description and references
D1/D2 dopaminergic agonist that also decreases plasma prolactin concentrations. Prepn: E. C. Kornfeld, N. J. Bach, US 4166182 (1979 to Lilly). Industrial synthesis: W. Cabri et al., Org. Process Res. Dev. 10, 198 (2006). Dopaminergic effects in rats: R. W. Fuller et al., Life Sci. 24, 375 (1979); T. T. Yen et al., ibid. 25, 209 (1979). Clinical pharmacology: L. Lemberger, R. E. Crabtree, Science 205, 1151 (1979). Pharmacological evaluation as antiparkinson agent: W. C. Koller, Neuropharmacology 19, 831 (1980). Clinical study in galactorrhea: J. T. Callaghan et al., Life Sci. 28, 95 (1981); in hyperprolactinemia: S. Francks et al., Lancet 2, 659 (1981); in treatment of pituitary tumors secreting prolactin or growth hormone: D. L. Kleinberg et al., N. Engl. J. Med. 309, 704 (1983). Clinical studies in Parkinson's disease: J. Jankovic, J. Orman, Adv. Neurol. 45, 551 (1986); C. W. Olanow, M. J. Alberts, ibid. 555; in restless legs syndrome: C. Trenkwalder et al., Neurology 62, 1391 (2004). Comprehensive description: D. J. Sprankle, E. C. Jensen, Anal. Profiles Drug Subs. Excip. 21, 375-413 (1992). Review of clinical experience in Parkinson's disease: U. Bonuccelli et al., Clin. Neuropharmacol. 25, 1-10 (2002).
Properties
Solid, mp 217.5°.Derivative
Mesylate.Nomenclature
Properties
Crystals, mp 258-260° (dec) (Sprankle, Jensen); also reported as mp 267.4° (Cabri). uv max (water): 279 nm (ε 6385); (methanol): 280 nm (ε 6980); (dehydrated ethanol): 281 nm (ε 6993). αD20 between -18.0° and -23.0° (c = 10 mg/ml in DMF). pKa (66% DMF) 7.8. Sparingly sol in DMF, methanol; slightly sol in water, 0.01N HCl, chloroform, acetonitrile, dichloromethane, dehydrated ethanol; very slightly sol in acetone. Practically insol in 0.1N NaOH, 0.1N HCl, ether. Partition coefficient at 25° (chloroform/water): 6.14 (pH 2.19); 119.6 (pH 4.02).Therapeutic Category
Antiparkinsonian.
Therapeutic Category (Veterinary)
In treatment of equine Cushing's disease.Keywords
Antiparkinsonian; Dopamine Receptor Agonist