7169. Perimycin
Nomenclature
Description and references
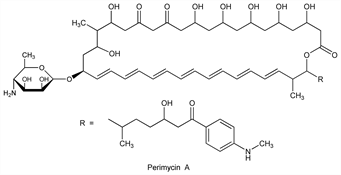
Polyene antifungal antibiotic complex produced by Streptomyces coelicolor var. aminophilus NRRL 2390: W. E. Wooldridge, US 2956925 (1960); R. R. Mohan et al., Antimicrob. Agents Chemother. 1963, 462; L. E. McDaniel et al., US 3182004 (1965 to Warner-Lambert). Purification: E. Borowski et al., Antimicrob. Agents Annu. 1960, 532. Found to be a mixture of three active components, perimycin A (major), B, and C. Mechanism of action with succinyl adduct: E. Borowski, B. Cybulska, Nature 213, 1034 (1967). Ionophoric and hemolytic activities: B. Cybulska et al., Biochem. Pharmacol. 38, 1755 (1989). Isoln of perimycins A, B, C and structural elucidation of A: P. Kolodziejczyk et al., Tetrahedron Lett. 1976, 3603. Stereochemistry and revised structure: J. Pawlak et al., J. Antibiot. 48, 1034 (1995).
Properties
Amorphous, golden-yellow solid. Has no definite mp but dec slowly with darkening upon heating. uv max (methanol): 383 nm (E1%1cm 1000). Sol in the following solvents in the presence of water: lower alcohols, pyridine, tetrahydrofuran, acetone, dioxane. Sol in warm methanol, DMF, dimethylsulfoxide, and in the lower fatty acids. Practically insol in water, petr ether, ethyl acetate, benzene.Derivative
Perimycin A.Nomenclature
Properties
uv max: 380 nm (E11 1000).Therapeutic Category
Antifungal.
Keywords
Antifungal (Antibiotics); Polyenes