7170. Perindopril
Nomenclature
Description and references
Angiotensin-converting enzyme (ACE) inhibitor. Hydrolyzed in vivo to the active diacid metabolite. Prepn: G. Remond et al., EP 49658 (1982 to Sci. Union et Cie. - Soc. Franc. Rech. Med.); M. Vincent et al., US 4508729 (1985 to ADIR). Stereoselective synthesis: M. Vincent et al., Tetrahedron Lett. 23, 1677 (1982). NMR study: N. Platzer et al., Magn. Reson. Chem. 26, 296 (1988). Hemodynamic effects in humans: K. R. Lees, J. L. Reid, Br. J. Clin. Pharmacol. 23, 159 (1987). Clinical evaluation in essential hypertension: T. Morgan et al., J. Cardiovasc. Pharmacol. 10, Suppl. 7, S116 (1987). Pharmacokinetics, pharmacodynamics of the diacid: K. R. Lees, J. L. Reid, ibid. 10, 129 (1987). Review of pharmacology and clinical use: P. A. Todd, A. Fitton, Drugs 42, 90-114 (1991). Symposium on clinical efficacy in cardiovascular disease: Am. J. Cardiol. 88, Suppl. 1, 1-40 (2001).
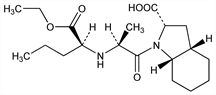
Derivative
tert-Butylamine.Nomenclature
Derivative
Diacid form.Nomenclature
Therapeutic Category
Antihypertensive.
Keywords
ACE-Inhibitor; Antihypertensive; N-Carboxyalkyl (peptide/lactam) Derivatives