7180. Permethrin
Nomenclature
Description and references
Synthetic pyrethroid insecticide, more stable to light and at least as active as the natural pyrethrins and with low mammalian toxicity: M. Elliott et al., Nature 246, 169 (1973). Of the four possible isomers, the (1R,trans)- and the (1R,cis)-isomers are the two esters primarily responsible for insecticidal activity: P. E. Burt et al., Pestic. Sci. 5, 791 (1974). Prepn of the racemic mixture: T. Mizutani et al., DE 2437882 (1975 to Sumitomo); F. Mori et al., DE 2544150; eidem, US 4113968 (1976, 1978, both to Kuraray). Metabolism: M. Elliott et al., J. Agric. Food Chem. 24, 270 (1976); L. C. Gaughan et al., ibid. 26, 613 (1978). Photodecompn: R. L. Holmstead et al., ibid. 590. Analysis: H. Swaine, M. J. Tandy, Anal. Methods Pestic. Plant Growth Regul. 13, 103-120 (1984). Field evaluation to control flies and ticks in cows: S. S. Quisenberry, D. R. Strohbehn, J. Econ. Entomol. 77, 422 (1984); R. B. Davey, E. H. Ahrens, Am. J. Vet. Res. 45, 1008 (1984). Clinical trial in pediatric pediculosis: E. Ares Mazas et al., Int. J. Dermatol. 24, 603 (1985). Toxicity studies in mammals: L. Metker et al., U.S. NTIS AD Rep. 1977, AD-AO47284, 70 pp; F. Cantalamessa, Arch. Toxicol. 67, 510 (1993). Review: C. N. E. Ruscoe, Pestic. Sci. 8, 236 (1977). Review of toxicology and human exposure: Toxicological Profile for Pyrethrins and Pyrethroids (PB2004-100004, 2003) 332 pp.
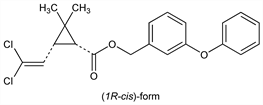
Properties
Technical material is a mixture of ≈60% trans- and 40% cis-isomers: Colorless crystals to a pale yellow viscous liquid, mp ≈35°. bp0.05 220°. d20 1.190-1.272. Vapor pressure at 50° <1 × 10-6 mm Hg. Soly in water: <1 ppm. Sol or miscible with org solvents except ethylene glycol. LD50 orally in female rats: 3801 mg/kg (Metker). LD50 in 8 day old rats, male adult rats (mg/kg): 340.5, 1500.0 orally (Cantalamessa). Toxic to bees and fish.Caution
Mild irritant to skin and eyes (Metker).Use
Insecticide.Therapeutic Category
Ectoparasiticide.
Therapeutic Category (Veterinary)
Ectoparasiticide.Keywords
Ectoparasiticide