7235. Pheniramine
Nomenclature
CAS number: 86-21-5
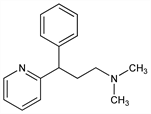
N,N-Dimethyl-γ-phenyl-2-pyridinepropanamine; 2-[α-(2-dimethylaminoethyl)benzyl]pyridine; 1-phenyl-1-(2-pyridyl)-3-dimethylaminopropane; 3-phenyl-3-(2-pyridyl)-N,N-dimethylpropylamine; prophenpyridamine; propheniramine. C16H20N2; mol wt 240.34.
C 79.96%, H 8.39%, N 11.66%.
Description and references
Synthesis: Sperber et al., US 2567245 and US 2676964 (1951, 1954, both to Schering).
Properties
Oily liq. Slightly yellow color. Characteristic amine-like odor. d 1.0081; bp13 181°; bp2 142°; bp0.5 135°; nD25 1.5519 to 1.5521. Insol in water. Sol in dil acids, alcohol, benzene, chloroform, ether.Derivative
Maleate.Nomenclature
CAS number: 132-20-7
Avil (Aventis); Daneral (Aventis); Inhiston (Biomed. Foscama); Trimeton (Schering). C16H20N2.C4H4O4; mol wt 356.42.
C 67.40%, H 6.79%, N 7.86%, O 17.96%.
Properties
Crystals from amyl alcohol, faint amine-like odor, mp 107°. Freely sol in water, alcohol. Slightly sol in ether, benzene. pH of 1% aq soln between 4.3 and 4.9.Derivative
p-Aminosalicylate.Nomenclature
CAS number: 3269-83-8
C16H20N2.C7H7NO3; mol wt 393.48.
C 70.21%, H 6.92%, N 10.68%, O 12.20%.
Properties
Small needles from acetone + ethyl acetate, large octahedra from water, dec 142°. One gram dissolves in 10 ml water, freely sol in alcohol. Sparingly sol in ethyl acetate, ether, acetone.Therapeutic Category
Antihistaminic.
Keywords
Antihistaminic; Alkylamine Derivatives