7277. Phenylbutazone
Nomenclature
Description and references
Prepn: Stenzl, US 2562830 (1951 to Geigy); cf. GB 812449 (1959 to Geigy). Review of synthesis: Ullmanns Encyklop"adie der technischen Chemie vol. 13, 298 (1962). Physical properties and pharmacology: v. Rechenberg, Phenylbutazone (Edward Arnold, London, 1962) 197 pp. Acute toxicity: T. B. Gaines, R. E. Linder, Fundam. Appl. Toxicol. 7, 299 (1986). Soly data: Pulver et al., Schweiz. Med. Wochenschr. 86, 1080 (1956). Comprehensive description: S. L. Ali, Anal. Profiles Drug Subs. 11, 483-521 (1982). Review of hematological effects: G. A. Faich, Pharmacotherapy 7, 25 (1987).
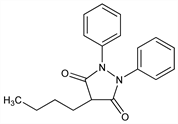
Properties
Crystals from ethanol, mp 105°. Soly in water at 22.5°: 0.7 mg/ml (also reported as 2.2 mg/ml). pK 4.5 (from uv in water), pK 4.89 (titration in 50% ethanol), pK 5.25 (titration in 80% 2-methoxyethanol). uv max (acid methanol): 239.5 nm (log ε 4.19).Derivative
Sodium salt.Nomenclature
Properties
Crystals, freely sol in water. pH of aq solns ≈8.2.Derivative
Calcium salt.Nomenclature
Ticinil Calcico (De Angeli).Properties
LD50 in adult male, female rats (mg/kg): 1311, 647 orally (Gaines, Linder).Derivative
Piperazine salt.Nomenclature
Pyrazinobutazone; pyrasanone; Carudol (Fher).Properties
mp 140-141° (solidifies and remelts at ≈180°).Derivative
2-Amino-2-thiazoline salt.Nomenclature
Description and references
Prepn: J. Moragues et al., Arzneim.-Forsch. 24, 1785 (1974). Pharmacology: M. Márquez, D. J. Roberts, ibid. 1786, 1790; M. Colombo et al., ibid. 26, 1347 (1976).
Properties
White crystals, mp 164-166°. LD50 orally in rats, mice: 1425, 1650 mg/kg (Colombo).Therapeutic Category
Anti-inflammatory.
Therapeutic Category (Veterinary)
Analgesic; anti-inflammatory.Keywords
Anti-inflammatory (Nonsteroidal); Pyrazolones