7304. o-Phenylphenol
Nomenclature
CAS number: 90-43-7
(1,1′-Biphenyl)-2-ol; 2-biphenylol; orthoxenol; o-hydroxydiphenyl; 2-hydroxydiphenyl; Dowicide 1 (Dow). C12H10O; mol wt 170.21.
C 84.68%, H 5.92%, O 9.40%.
Description and references
Prepn from phenyl ether: Lüttringhaus, S"a"af, Ann. 542, 241 (1939); from dibenzofuran: Gilman, Esmay, J. Am. Chem. Soc. 75, 2947 (1953); Müller, US 2862035 (1958 to Bayer). Purification: Widiger, US 3087969 (1963 to Dow). Toxicity data: Hodge et al., J. Pharmacol. Exp. Ther. 104, 202 (1952). Review of carcinogenic risk: IARC Monographs 30, 329-344 (1983).
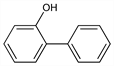
Properties
White, flaky crystals. Mild, characteristic odor. mp 55.5-57.5°; bp 280-284°; bp15 152-154°. Practically insol in water. Sol in fixed alkali hydroxide solns and most organic solvents. LD50 orally in rats: 2.48 g/kg (Hodge).Derivative
Sodium salt.Nomenclature
CAS number: 132-27-4
Natriphene; sodium o-phenylphenate; sodium 2-phenylphenoxide; Dowicide A (Dow). C12H9NaO; mol wt 192.19.
C 74.99%, H 4.72%, Na 11.96%, O 8.32%.