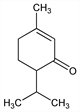
7473. Piperitone
Nomenclature
CAS number: 89-81-6
3-Methyl-6-(1-methylethyl)-2-cyclohexen-1-one; p-menth-1-en-3-one; 4-isopropyl-1-methyl-1-cyclohexen-3-one. C10H16O; mol wt 152.23.
C 78.90%, H 10.59%, O 10.51%.
Description and references
Isoln of d-form from oil of Cymbopogon sennaarensis Chiov., Gramineae: Roberts, J. Chem. Soc. 107, 1465 (1915); from oil of Andropogon iwarancusa Jones: Simonsen, ibid. 119, 1644 (1921); from oil of Mentha spp., Labiatae: Reitsma, J. Am. Pharm. Assoc. 47, 265, 267 (1958). Isoln of l-form from Sitka spruce oil: von Rudloff, Can. J. Chem. 42, 1057 (1964). Isoln of dl-form from oil of Eucalyptus dives Schau., Myrtaceae: Read, Smith, J. Chem. Soc. 119, 779 (1921); from peppermint oil: Katsuragi, Koryo No. 24, 16 (1953). Synthesis of dl-form: Misrock, Church, Ind. Eng. Chem. 49, 822 (1957); Bain et al., US 2972632 (1961 to Glidden); Wiemann, Dubois, Bull. Soc. Chim. Fr. 1962, 1813; Stepanov, Myrsina, Zh. Obshch. Khim. 34, 3092 (1964).