7512. Pivampicillin
Nomenclature
CAS number: 33817-20-8
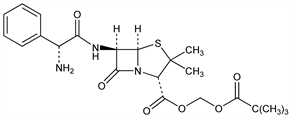
(2S,5R,6R)-6-[[(2R)-Aminophenylacetyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid (2,2-dimethyl-1-oxopropoxy)methyl ester; hydroxymethyl 6-(2-amino-2-phenylacetamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate
pivalate (ester); 6-[d-α-aminophenylacetamido]penicillanic acid pivaloyloxymethyl
ester; pivaloyloxymethyl d-α-aminobenzylpenicillinate; ampicillin pivaloyloxymethyl ester; pivaloyloxymethyl ampicillinate; MK-191. C22H29N3O6S; mol wt 463.55.
C 57.00%, H 6.31%, N 9.06%, O 20.71%, S 6.92%.
Description and references
Semi-synthetic antibiotic related to penicillin. Prepn: E. K. Frederiksen, W. O. Godtfredsen, ZA 6805952; eidem, US 3660575 (1969, 1972 to L"ovens Kemiske Fabrik); von Daehne et al., J. Med. Chem. 13, 607 (1970). Pharmacology: eidem, loc. cit.; Jordan et al., Antimicrob. Agents Chemother. 1970, 438; Foltz et al., ibid. 442. Pharmacokinetics in man: M. Ehrnebo et al., J. Pharmacokinet. Biopharm. 7, 429 (1979). Toxicity: von Daehne et al., Antimicrob. Agents Chemother. 1970, 430.
Derivative
Hydrochloride.Nomenclature
CAS number: 26309-95-5
Pondocil (Leo Pharm); Pondocillin (Leo Pharm); Pondocillina (Sigma-Tau); Sanguicillin (Zdravlje). C22H29N3O6S.HCl; mol wt 500.01.
C 52.85%, H 6.05%, N 8.40%, O 19.20%, S 6.41%, Cl 7.09%.
Properties
Microcrystalline powder, mp 155-156° (decomp). [α]D20 +196° (c = 1 in water). Weak uv max (water): 268, 262, 256 nm (E1%1cm ≈3.9, 5.7, 6.3). pKa ≈7.0. Relatively stable in acid soln; ester hydrolyzes slowly in neutral soln. pH of 0.5 g/100 ml water: ≈4.5. Very sol in water and chloroform; freely sol in ethanol; sparingly sol in n-propanol, tert-butanol, and ethyl ether. LD50 in mice, rats (g/kg): 3.34, 5.00 orally; 3.60, 4.50 s.c. (von Daehne).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins