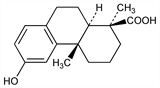
7543. Podocarpic Acid
Nomenclature
Description and references
Chief acidic constituent of the resin of the Javanese Podocarpus cupressina (L'Hérit.) Pers., Coniferae, esp. in var. imbricata. Also in New Zealand kahikatea resin (from Podocarpus dacrydioides) and in rimu resin (Dacrydium cupressinum). A related component, nimbiol, q.v., occurs in the nim tree, Azadirachta indica Juss. (Melia azadirachta L.), Meliaceae. Isoln from Podocarpus cupressina: A. C. Oudemans, Ber. 6, 1122 (1873); from Dacrydium cupressinum: I. R. Sherwood, W. F. Short, J. Chem. Soc. 1938, 1008. Structure: Campbell, Todd, J. Am. Chem. Soc. 64, 928 (1942). Conversion to nimbiol as proof of structure: Bible, Tetrahedron 11, 22 (1960). Synthesis from desoxypodocarpic acid: Wenkert, Jackson, J. Am. Chem. Soc. 80, 217 (1958). Total synthesis of dl-form: Meyer, Maheshwari, Tetrahedron Lett. 1964, 2175; P. R. Kanjilal et al., Synth. Commun. 11, 795 (1981). Synthesis: Pelletier et al., Tetrahedron Lett. 1971, 4179.