7573. Polymyxin
Nomenclature
Description and references
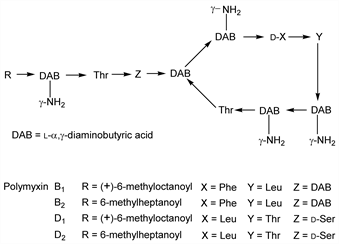
Antibiotic complex produced by Bacillus polymyxa: Brownlee, Jones, Biochem. J. 43, XXV (1948). Prepn: Ainsworth, Pope, US 2565057 (1951 to Burroughs Wellcome); Petty, US 2595605 (1952 to Am. Cyanamid); Benedict, Stodola, US 2771397 (1956 to USDA). Purification: Hastings et al., GB 782926 (1957 to Distillers Co.). Polymyxins A, B, C, D, E, F, K, M, P, S and T have been identified. Isoln of polymyxins A, B, C and E: Few, Schulman, Biochem. J. 54, 171 (1953); of polymyxin D: Stansley et al., Bull. Johns Hopkins Hosp. 81, 43 (1947); of polymyxin F: W. L. Parker et al, J. Antibiot. 30, 767 (1977); of polymyxin K: Kimura, JP 71 16152 (1971), C.A. 75, 62180r (1971); of polymyxin M: Khokhlov et al., C.A. 55, 5653h (1961); of polymyxin P: Kimura et al., J. Antibiot. 22, 449 (1969); of polymyxin S1 and polymyxin T1: J. Shoji et al., ibid. 30, 1029 (1977). Resolution of polymyxin B into B1 and B2: Hausmann, Craig, J. Am. Chem. Soc. 76, 4892 (1954). Structure and synthesis of polymyxin B1: Wilkinson, Lowe, Nature 202, 1211 (1964); Vogler et al., Helv. Chim. Acta 48, 1161 (1965). Structure of polymyxin B2: Wilkinson, Lowe, Nature 204, 993 (1964). Separation of polymyxin D into D1 and D2 and structures: Hayashi et al., Experientia 22, 354 (1966). Structure of polymyxin S1: J. Shoji et al., J. Antibiot. 30, 1035 (1977); of polymyxin T1: eidem, ibid. 1042. Review: Vogler, Studer, Experientia 22, 345-354 (1966); Paulus, “Polymyxins” in Antibiotics II, D. Gottlieb, P. Shaw, Eds. (Springer-Verlag, New York, 1967) pp 254-267.
Properties
Obtained as the hydrochloride. Nearly colorless powder, dec 228-230°. [α]D23 -40° (c = 1.05). The hydrochloride is very sol (more than 40%) in water and methanol. The soly decreases in the higher alcs. Practically insol in the usual ethers, esters, ketones, hydrocarbons, and the chlorinated solvents. Forms water insol salts with a number of ppts such as picric acid, helianthic acid, Reinecke salt. The free base is slightly sol in water; almost insol in alc.Derivative
Polymyxin B.Nomenclature
Properties
Mixture of polymyxins B1 and B2. [α]5461 -106.3° (1N HCl).Derivative
Polymyxin B sulfate.Nomenclature
Description and references
Solubilities: Weiss et al., Antibiot. Chemother. 7, 374 (1957).
Derivative
Polymyxin B sulfate mixture with trimethoprim.Nomenclature
Polytrim (Wellcome).Derivative
Polymyxin B1.Nomenclature
Derivative
Polymyxin B1 pentahydrochloride.Properties
White powder. [α]D25 -85.11° (c = 2.33 in 75% ethanol).Derivative
Polymyxin B2.Nomenclature
Properties
[α]225461 -112.4° (2% Acetic acid).Derivative
Polymyxin D1.Nomenclature
Derivative
Polymyxin D2.Nomenclature
Derivative
Polymyxin E see Colistin.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Polypeptides