7576. Polyoxins
Nomenclature
Description and references
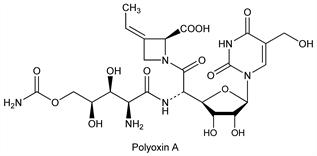
Agricultural antifungal antibiotic complex produced by Streptomyces cacaoi var asoensis and S. piomogenus. Polyoxins A through O are known, all except C and I having specific activity against phytopathogenic fungi by inhibiting cell wall chitin synthesis. Isoln and characterization of polyoxin A: S. Suzuki et al., J. Antibiot. 18A, 131 (1965); of A and B: K. Isono et al., Agric. Biol. Chem. 29, 848 (1965); S. Suzuki et al., JP 66 15520; of C through L: K. Isono et al., Agric. Biol. Chem. 30, 817 (1966); ibid. 31, 190 (1967); ibid. 32, 792 (1968); of M: K. Isono, S. Suzuki, Tetrahedron Lett. 1970, 425. Isoln of polyoxins N and O: S. Suzuki et al., JP Kokai 72 23596 (to Inst. Phys. Chem. Res., and Hokko Chem. Ind.), C.A. 78, 41566t (1973); production of N and O: eidem, JP 77 20555 (1977 to Inst. Phys. Chem. Res.), C.A. 87, 150183x (1977). Isoln and structure of N: M. Uramoto et al., Nucleic Acids Res. Spec. Publ. 5, 327 (1978). Structural elucidation of polyoxins A through L: K. Isono et al., J. Am. Chem. Soc. 91, 7490 (1969) DOI PubMed. Revised structure of A: S. Hanessian et al., Tetrahedron Lett. 34, 4153 (1993) DOI. Total synthesis of J: H. Kuzuhara et al., ibid. 1973, 5055. Biosynthetic studies: K. Isono et al., Biochemistry 14, 2992 (1975) DOI PubMed; S. Funayama, K. Isono, ibid. 5568; K. Isono, R. J. Suhadolnick, Arch. Biochem. Biophys. 173, 141 (1976) DOI PubMed; S. Funayama, K. Isono, Biochemistry 16, 3121 (1977) DOI PubMed; K. Isono et al., J. Am. Chem. Soc. 100, 3937 (1978) DOI. Mode of action: N. Ohta et al., Agric. Biol. Chem. 34, 1224 (1970); M. Hori et al., ibid. 35, 1280 (1971); 38, 691, 699 (1974). Review: R. J. Suhadolnick, Nucleoside Antibiotics (Wiley-Interscience, New York, 1970) pp 218-234; K. Isono, S. Suzuki, Heterocycles 13, 333-351 (1979).