7703. Pralidoxime Chloride
Nomenclature
Description and references
Cholinesterase reactivator. Prepn of salts: I. B. Wilson et al., US 2816113 (1957 to U.S. Sec'y. of Army); A. A. Kondritzer et al., J. Pharm. Sci. 50, 109 (1961). Manufacturing processes: R. I. Ellin et al., US 3140289 (1964 to U.S. Dept. of the Army); McDowell, US 3155674 (1964 to Olin Mathieson). Commercial prepn: R. I. Ellin, Ind. Eng. Chem. Prod. Res. Dev. 3 (1), 20 (1964). HPLC determn in serum: P. Houzé et al., J. Chromatogr. B 814, 149 (2005). Pharmacokinetics: D. Jovanovic, Arch. Toxicol. 63, 416 (1989). Toxicology: Christensen, Richter, Arch. Environ. Health 15, 599 (1967). Toxicity data: Fleisher et al., Toxicol. Appl. Pharmacol. 16, 40 (1970); R. I. Ellin, J. H. Wills, J. Pharm. Sci. 53, 1143 (1964). Comprehensive description: U. V. Banakar, U. N. Patel, Anal. Profiles Drug Subs. 17, 533-569 (1988). Clinical trial in organophosphorus pesticide poisoning: K. S. Pawar et al., Lancet 368, 2136 (2006).
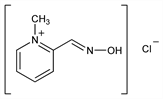
Properties
Crystals from alcohol + ether, mp 235-238° (dec). Soly (g/100 ml) 25°: acetone 0, isopropanol 0.09, ethanol 0.89, methanol 8.5, water 65.5. LD50 in rats (mg/kg): 96 i.v. (Fleisher). LD50 in rabbits (mg/kg): 95 i.v.; LD50 in mice (mg/kg): 115 i.v., 205 i.p., 4100 orally (Ellin, Wills).Derivative
Pralidoxime iodide.Nomenclature
Properties
Yellow crystals from alcohol, mp 225-226°. Soly at 25°: 48 mg/ml. Very sol in water, fairly sol in hot alcohol, poorly sol in cold alcohol. Insol in ether, acetone. LD50 in mice (mg/kg): 140-178 i.v., 136-260 i.p., 290-340 s.c., 1500-4000 orally (Ellin, Wills).Derivative
Pralidoxime mesylate.Nomenclature
Properties
Very hygroscopic crystals from ethanol, mp 155°. pKa 8.0. Soly in water: 1 g in 2 ml. LD50 in mice (mg/kg): 118-122 i.v., 216 i.p., 3700 orally; in rats (mg/kg): 109 i.v., 262 i.p. (Ellis, Wills).Therapeutic Category
Antidote (nerve gases and organophosphate insecticide poisoning).
Therapeutic Category (Veterinary)
Antidote (organophosphate poisoning).Keywords
Cholinesterase Reactivator; Antidote (Organophosphate Poisoning)