7721. Prednisolone
Nomenclature
Description and references
Synthetic corticosteroid; metabolically interconvertible with prednisone, q.v. Prepn: A. Nobile et al., J. Am. Chem. Soc. 77, 4184 (1955); A. Nobile, US 2837464; US 3134718 (1958, 1964, both to Schering); Herzog et al., Tetrahedron 18, 581 (1962). HPLC determn in urine: B. M. Frey, F. J. Frey, J. Chromatogr. 229, 283 (1982). Clinical use in advanced cancer: R. G. Twycross, D. Guppy, Practitioner 229, 57 (1985); in inflammatory bowel disease: C. J. Hawkey, Neth. J. Med. 35 Suppl 1, S21-S26 (1989). Review of clinical pharmacology: W. J. Jusko, J. Q. Rose, Ther. Drug Monit. 2, 169-176 (1980); of pharmacokinetics and metabolism: B. M. Frey, F. J. Frey, Clin. Pharmacokinet. 19, 126-146 (1990). Comprehensive description: S. L. Ali, Anal. Profiles Drug Subs. Excip. 21, 415-500 (1992).
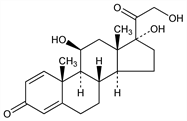
Properties
Crystals, dec 240-241°. [α]D25 +102° (dioxane). uv max (methanol): 242 nm (ε 15000; A1%1cm 414). Very slightly sol in water. One gram dissolves in about 30 ml of alcohol, in about 180 ml of chloroform, in about 50 ml of acetone. Sol in methanol, dioxane.Derivative
21-Acetate.Nomenclature
Properties
Crystals, decomp 237-239°. [α]D25 +116° (dioxane).Derivative
21-tert-Butylacetate.Nomenclature
Description and references
Prepn: Sarett, US 2736734 (1956 to Merck & Co.).
Properties
Crystals from ethanol, mp 266-273°.Derivative
21-Disodium Phosphate.Nomenclature
Description and references
Prepn: Sarett, US 2789117 (1957 to Merck & Co.). Alternate synthesis: Poos et al., Chem. Ind. (London) 1958, 1260; Elks, Phillips, US 2936313 (1960 to Glaxo).
Properties
White powder. Slightly hygroscopic. Stable at room temp. [α]D25 +102.5°. uv max (methanol): 243 nm (A1%1cm 308). Sol in water, methanol, ethanol. pH of 1% aq soln 7.5 to 8.5.Derivative
21-Succinate sodium salt.Nomenclature
Description and references
Prepn: Shull, Kita, DE 1045400 (1958 to Pfizer), C.A. 55, 2746f (1961).
Derivative
21-Stearoylglycolate.Nomenclature
Description and references
Prepn: Giraldi, Nannini, US 3171846 (1965 to Carlo Erba); Giraldi et al., Arzneim.-Forsch. 16, 162 (1966).
Properties
Crystals from dilute alcohol or butyl ether, mp 105-107°. uv max (methanol): 242 nm (E1%1cm 212 ±10). [α]D20 +57-63°.Derivative
21-m-Sulfobenzoate sodium salt.Nomenclature
Description and references
Prepn: Allais, Girault, US 3032568; Joly, Warnant, US 3037034 (both 1962 to Roussel-UCLAF).
Properties
Crystals from water, dec 293-295°. [α]D20 +170° (water).Derivative
21-Trimethylacetate.Nomenclature
Description and references
Prepn: Vischer et al., Helv. Chim. Acta 38, 1502 (1955); Joly, Warnant, US 3037034 (1962 to Roussel-UCLAF).
Properties
Crystals from acetone, mp 233-236°. [α]D26 +103° (c = 1.208 in chloroform); [α]D20 +97.5° (c = 1 in chloroform). uv max: 244 nm (ε 14700).Therapeutic Category
Glucocorticoid.
Therapeutic Category (Veterinary)
Glucocorticoid.Keywords
Glucocorticoid