7722. Prednisone
Nomenclature
CAS number: 53-03-2
17,21-Dihydroxypregna-1,4-diene-3,11,20-trione; 1,4-pregnadiene-17α,21-diol-3,11,20-trione; Δ1-dehydrocortisone; Δ1-cortisone; deltacortisone; delta E; metacortandracin; retrocortine; NSC-10023; Colisone (Frosst); Cortancyl (Aventis); Dacortin (Merck KGaA); Decortin (Merck KGaA); Deltacortene (Bruno); Deltacortone (Merck & Co.); Deltasone (Upjohn); Deltison (Ferring); Di-Adreson (Organon); Encorton (Polfa); Hostacortin (Hoechst); Meticorten (Schering-Plough); Orasone (Solvay); Rectodelt (Trommsdorff); Sone (Fawns & McAllan); Ultracorten (Novartis). C21H26O5; mol wt 358.43.
C 70.37%, H 7.31%, O 22.32%.
Description and references
Prepn: Oliveto, Gould, US 2897216 (1959 to Schering). Microbiological prepn: Nobile et al., J. Am. Chem. Soc. 77, 4184 (1955); Nobile, US 2837464 and US 3134718 (1958, 1964 both to Schering); Herzog et al., Tetrahedron 18, 581 (1962). Structure: Herzog et al., Science 121, 176 (1955); cf. Djerassi et al., US 2579479 (1951 to Syntex).
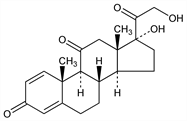
Properties
Crystals, dec 233-235°. [α]D25 +172° (dioxane). uv max (methanol): 238 nm (ε 15500). Very slightly sol in water. One gram dissolves in about 150 ml alcohol, in about 200 ml chloroform. Slightly sol in methanol, dioxane.Derivative
21-Acetate.Nomenclature
CAS number: 125-10-0
C23H28O6; mol wt 400.46.
C 68.98%, H 7.05%, O 23.97%.
Properties
Crystals, dec 226-232°. [α]D25 +186° (dioxane). uv max (ethanol): 238 nm (ε 16100).Therapeutic Category
Glucocorticoid.
Therapeutic Category (Veterinary)
Adrenocortical steroid. Glucocorticoid, anti-inflammatory.Keywords
Glucocorticoid