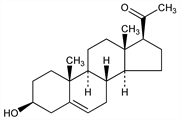
7733. Pregnenolone
Nomenclature
CAS number: 145-13-1
(3β)-3-Hydroxypregn-5-en-20-one; Δ5-pregnen-3β-ol-20-one; 17β-(1-ketoethyl)-Δ5-androsten-3β-ol. C21H32O2; mol wt 316.48.
C 79.70%, H 10.19%, O 10.11%.
Description and references
Prepn from stigmasterol: Butenandt et al., Ber. 67, 1611 (1934); Butenandt, Fleischer, Ber. 70, 96 (1937); from androstenolone: H. Butenandt, J. Schmidt-thome, Ber. 72, 182 (1939); S. Danishefsky et al., J. Org. Chem. 40, 1989 (1975); from Δ5-3-acetoxyetiocholenic acid chloride: Wettstein, Helv. Chim. Acta 23, 1373 (1940); from diosgenin: Marker, Krueger, J. Am. Chem. Soc. 62, 3349 (1940); Marker et al., ibid. 69, 2173 (1947); from nologenin: eidem, ibid. 2395; by treating a 21-halo-Δ5-pregnen-3-ol-20-one with a reducing agent: CH 215139 (1941), C.A. 42, 3144 (1948). Crystal structure: J. Bordner et al., Cryst. Struct. Commun. 7, 513 (1978).
Properties
Needles from dil alc, mp 193°. [α]D20 +28° (alc). Very sparingly sol in water. Soly (g/100 ml of soln): carbon tetrachloride 0.5; petr ether 0.1; ethyl acetate 1.1; acetone 0.6; chloroform 17.0; ethanol 1.9; benzene 0.9; isopropanol 1.5. Soly (g/100 ml of solvent): propylene glycol 0.1; dioxane 3.1; benzyl alcohol 8.1. On refluxing with methyl alcohol yields the 17-isopregnenolone, mp 172-173°, [α]D20 -140.5° (alcohol).Derivative
Acetate.C23H34O3; mol wt 358.51.
C 77.05%, H 9.56%, O 13.39%.
Properties
Needles from alcohol, mp 149-151°. [α]D20 +22° (alcohol). Soly (g/100 ml of soln): carbon tetrachloride 5.0; petr ether 1.0; ethyl acetate 7.9; acetone 2.7; chloroform 55.0; ethanol 2.5; benzene 26.0; isopropanol 2.0. Soly (g/100 ml of solvent): propylene glycol 0.1; dioxane 20.2; benzyl alcohol 11.1; benzyl benzoate 9.1.Derivative
Methyl ether.Nomenclature
CAS number: 511-26-2
C22H34O2; mol wt 330.50.
C 79.95%, H 10.37%, O 9.68%.