7732. 4-Pregnene-17α,20β,21-triol-3-one
Nomenclature
CAS number: 128-19-8
(20R)-17,20,21-Trihydroxypregn-4-en-3-one; 17-(1,2-dihydroxyethyl)-Δ4-androsten-3-on-17α-ol; 17α-pregnenetriolone. C21H32O4; mol wt 348.48.
C 72.38%, H 9.26%, O 18.36%.
Description and references
Prepd from 17-ethynyltestosterone by hydrogenation with palladium in pyridine, allylic rearrangement, and hydroxylation with osmium tetroxide: Ruzicka, Müller, Helv. Chim. Acta 22, 755 (1939); Logemann, Naturwissenschaften 27, 196 (1939); from a 3-enol ester of a 17,21-diacyloxyprogesterone by reduction followed by saponification: CH 207496 (1940), C.A. 36, 3636 (1942).
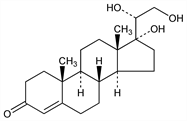
Properties
Crystals from methanol. mp 190°. Sol in dioxane, chloroform, methanol. [α]D +63° (c = 1 in dioxane). uv max: 240 nm (log ε 4.1).Derivative
20,21-Diacetate.C25H36O6; mol wt 432.55.
C 69.42%, H 8.39%, O 22.19%.