7757. Procaine
Nomenclature
CAS number: 59-46-1
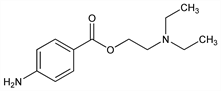
4-Aminobenzoic acid 2-(diethylamino)ethyl ester; p-aminobenzoyldiethylaminoethanol; 2-diethylaminoethyl p-aminobenzoate. C13H20N2O2; mol wt 236.31.
C 66.07%, H 8.53%, N 11.85%, O 13.54%.
Description and references
Benzoic acid derivative with anesthetic activity. Prepn: A. Einhorn, US 812554 (1906); idem, Ann. 371, 125 (1909); A. Einhorn, E. Uhlfelder, ibid. 131. CNS effects: C. G. Peterson, Anesthesiology 16, 976 (1955). Intravenous pharmacokinetics in humans: A. B. Seifen et al., Anesth. Analg. 58, 382 (1979). Clinical evaluation as anti-arrhythmic and cough suppressant during anesthesia: D. S. Thompson et al., Am. J. Surg. 138, 798 (1979). Stabilization of vascular smooth muscle in vitro: K. Kitamura et al., Drugs Exp. Clin. Res. 12, 773 (1986). Toxicity data: W. C. North, K. F. Urbach, J. Am. Pharm. Assoc. Sci. Ed. 45, 382 (1956); E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971).
Properties
Hygroscopic, anhydr plates, tablets from ligroin or ether, mp 61°. When freshly precipitated, one gram dissolves in 200 ml water. Sol in alc, ether, benzene, chloroform. LD50 in mice (mg/kg): 195 i.p.; 45 i.v. (North, Urbach).Derivative
Dihydrate.Nomenclature
CAS number: 6192-89-8
Properties
Needles from aq alc, mp 51°. Slightly bitter taste; applied to the tongue causes transitory numbing sensation.Derivative
Nitrate.Nomenclature
CAS number: 6192-92-3
C13H20N2O2.HNO3; mol wt 299.32.
C 52.16%, H 7.07%, N 14.04%, O 26.73%.
Properties
Crystals, mp 100-102°. Sol in water, alcohol. The aq soln is neutral. Particularly useful with silver nitrate because no precipitate forms.Derivative
Butyrate.Nomenclature
CAS number: 136-55-0
Probutylin (Rorer). C13H20N2O2.C4H8O2; mol wt 324.42.
C 62.94%, H 8.70%, N 8.63%, O 19.73%.
Properties
Hygroscopic crystals. Soluble in water, alcohol, vegetable oils.Derivative
Hydrochloride.Nomenclature
CAS number: 51-05-8
Anestil; Enpro (Kobayashi); Gero (URPAC); Jenacaine; Medaject (Neda); Naucaine; Neocaine; Novocain (Sanofi Winthrop); Omnicain (Daiichi); Planocaine (M & B); Rocain (Fuso); Syntocain (Sintetica). C13H20N2O2.HCl; mol wt 272.77.
C 57.24%, H 7.76%, N 10.27%, O 11.73%, Cl 13.00%.
Properties
Crystals. Six-sided plates, monoclinic or triclinic. mp 153-156°. Numbing taste. Stable in air. One gram dissolves in 1 ml water and in 30 ml alcohol. Slightly sol in chloroform. Almost insol in ether. The pH of a 0.1M aq soln is 6.0. Aq solns may be sterilized by boiling. LD50 in mice (mg/kg): 660 ± 60 s.c. (Goldenthal).Therapeutic Category
Anesthetic (local).
Therapeutic Category (Veterinary)
Anesthetic (local).Keywords
Anesthetic (Local)