7761. Prochlorperazine
Nomenclature
CAS number: 58-38-8
2-Chloro-10-[3-(4-methyl-1-piperazinyl)propyl]-10H-phenothiazine; 3-chloro-10-[3-(4-methyl-1-piperazinyl)propyl]phenothiazine; 2-chloro-10-[3-(1-methyl-4-piperazinyl)propyl]phenothiazine; N-[γ-(4′-methylpiperazinyl-1′)propyl]-3-chlorophenothiazine; chlormeprazine; prochlorpemazine; proclorperazine; Bayer A 173; RP-6140; SKF-4657. C20H24ClN3S; mol wt 373.94.
C 64.24%, H 6.47%, Cl 9.48%, N 11.24%, S 8.57%.
Description and references
Prepn: R. J. Horclois, GB 780193; FR 1167627; US 2902484 (1957, 1958, 1959 all to Rh^one-Poulenc). Pharmacology and toxicity: S. Courvoisier et al., C. R. Seances Soc. Biol. Ses Fil. 152, 1371 (1958). Pharmacokinetics: W. B. Taylor, D. N. Bateman, Br. J. Clin. Pharmacol. 23, 137 (1987). Clinical efficacy as antiemetic and antipsychotic: J. Lapierre et al., Can. Psychiatr. Assoc. J. 14, 267 (1969). Clinical trial in treatment of nausea, dizziness and vertigo: A. E. Ward, Br. J. Clin. Pract. 42, 228 (1988).
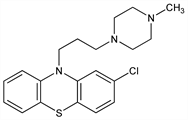
Derivative
Dimaleate.Nomenclature
CAS number: 84-02-6
Buccastem (Reckitt Benckiser); Compazine (GSK); Meterazine; Stemetil (Aventis); Vertigon (GSK). C20H24ClN3S.2C4H4O4; mol wt 606.09.
C 55.49%, H 5.32%, Cl 5.85%, N 6.93%, S 5.29%, O 21.12%.
Properties
Minute crystals, mp 228°. Very slightly sol in water (<0.1% at 20°). Slightly sol in methanol, ethanol. Practically insol in ether, benzene, chloroform. LD50 in mice (mg/kg): 400 s.c.; 120 i.p.; 90 i.v., 400 orally (Courvoisier).Derivative
Dimethanesulfonate.Nomenclature
CAS number: 51888-09-6
Prochlorperazine mesylate; Novamin (Shionogi); Tementil (Aventis). C20H24ClN3S.2CH4O3S; mol wt 566.15.
C 46.67%, H 5.70%, Cl 6.26%, N 7.42%, S 16.99%, O 16.96%.
Therapeutic Category
Antiemetic; antipsychotic. In treatment of vertigo.
Therapeutic Category (Veterinary)
Antiemetic.Keywords
Antiemetic; Antipsychotic; Phenothiazines