7763. Procyclidine
Nomenclature
CAS number: 77-37-2
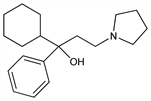
α-Cyclohexyl-α-phenyl-1-pyrrolidinepropanol; 1-cyclohexyl-1-phenyl-3-(1-pyrrolidinyl)-1-propanol; 1-cyclohexyl-1-phenyl-3-pyrrolidino-1-propanol. C19H29NO; mol wt 287.44.
C 79.39%, H 10.17%, N 4.87%, O 5.57%.
Description and references
Anticholinergic. Prepn of free base and hydrochloride: Adamson et al., J. Chem. Soc. 1951, 52; Adamson, US 2891890 (1959 to Burroughs Wellcome). Prepn of methochloride: Bottorff, US 2826590 (1958 to Lilly); Harfenist, Magnien, US 2842555 (1958 to Burroughs Wellcome).
Properties
Crystals from petr ether, mp 85.5-86.5°. uv max (0.17% in ethanol): 258.5 nm (ε 233).Derivative
Hydrochloride.Nomenclature
CAS number: 1508-76-5
Arpicolin (Rosemont); Kemadrin (Glaxo Wellcome); Osnervan (Glaxo Wellcome). C19H29NO.HCl; mol wt 323.90.
C 70.45%, H 9.34%, N 4.32%, O 4.94%, Cl 10.95%.
Properties
Crystals from ethanol + ethyl acetate, dec 226-227°. Moderately sol in water (about 3.0 g/100 ml); more sol in alcohol, chloroform; very slightly sol in ether.Derivative
Methochloride.Nomenclature
1-(3-Cyclohexyl-3-hydroxy-3-phenylpropyl)-1-methylpyrrolidinium chloride; tricyclamol chloride; Lergine.C19H29NO.CH3Cl; mol wt 337.93.
C 71.08%, H 9.54%, N 4.14%, O 4.73%, Cl 10.49%.
Properties
Crystals from nitroethane, mp 159-164°. Moderately sol in water, alcohol. Practically insol in ether.Derivative
Methosulfate.C21H35NO5S; mol wt 413.57.
C 60.99%, H 8.53%, N 3.39%, O 19.34%, S 7.75%.
Properties
Crystals, mp ≈100°. Soluble in water (about 2% at 25°), alcohol. Practically insol in ether.Therapeutic Category
Antiparkinsonian.
Keywords
Antiparkinsonian; Antimuscarinic