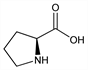
7780. Proline
Nomenclature
Description and references
Non-essential amino acid for human development. Only imino acid of the 20 amino acids commonly found in proteins. Shows cis-trans isomerism. First synthesized: R. Willst"atter, Ber. 33, 1160 (1900); prior to identification in casein: E. Fischer, ibid. 34, 454 (1901). Named in 1904 by Fischer. Early chemistry and biochemistry: Amino Acids and Proteins, D. M. Greenberg, Ed. (Charles C. Thomas, Springfield, IL, 1951) 950 pp., passim; J. P. Greenstein, M. Winitz, Chemistry of the Amino Acids vols 1-3 (John Wiley and Sons, Inc., New York, 1961) pp. 2178-2201, passim. Stereospecific synthesis: S. L. Titouani et al., Tetrahedron 36, 2961 (1980). HPLC determn: A. Carisano, J. Chromatogr. 318, 132 (1985). HPLC separation of cis-trans isomers of peptides: S. Friebe et al., J. Chromatogr. A 661, 7 (1994). Review of biosynthesis and degradation: E. Adams, L. Frank, Annu. Rev. Biochem. 49, 1005-1061 (1980). Review of effects on protein structure and biological function: A. Yaron, F. Naider, Crit. Rev. Biochem. Mol. Biol. 28, 31-81 (1993); G. Vanhoof et al., FASEB J. 9, 736-744 (1995).