7840. Propranolol
Nomenclature
Description and references
β-Adrenergic blocker. Prepn: BE 640312 and BE 640313; Crowther, Smith, US 3337628 and US 3520919 (1964, 1964, 1967, 1970 all to I.C.I.). Description of optical isomers: Howe, Shanks, Nature 210, 1336 (1966). Biological studies: Barrett, Cullum, Br. J. Pharmacol. 34, 43 (1968). Metabolism: Bond, Nature 213, 721 (1967). Multi-center clinical trial in myocardial infarction: V. Hansteen et al., Br. Med. J. 284, 155 (1982). CZE determn in urine: J. J. B. Nevado et al., Anal. Chim. Acta 559, 9 (2006). Use as migraine prophylactic: S. Diamond, E. Millstein, J. Clin. Pharmacol. 28, 193 (1988); S. Diamond et al., Headache 27, 70 (1987). Toxicity data: M. Martin, P. Linee, Eur. J. Med. Chem. 9, 563 (1974). Clinical evaluation in hypertension: D. A. Sica et al., J. Clin. Hypertens. 6, 231 (2004). Review of pharmacokinetics: P. A. Routledge, D. G. Shand, Appl. Pharmacokinet. 1980, 464-485; of pharmacology: J. D. Fitzgerald in Pharmacology of Antihypertensive Drugs, A. Scriabine, Ed. (Raven Press, New York, 1980) pp 195-208. Series of articles on use in hypertension: Drugs 37, Suppl. 2, 42-76 (1989).
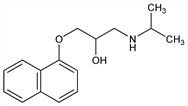
Properties
Crystals from cyclohexane, mp 96°.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from n-propanol, mp 163-164°. Sol in water, alc. Practically insol in ether, benzene, ethyl acetate. LD50 in mice (mg/kg): 565 orally; 22 i.v.; 107 i.p. (Martin, Linee).Therapeutic Category
Antihypertensive; antianginal; antiarrhythmic (class II).
Therapeutic Category (Veterinary)
Antiarrhythmic (class II).Keywords
β-Adrenergic Blocker; Antianginal; Antiarrhythmic; Antihypertensive; Aryloxypropanolamine Derivatives