7839. Propoxyphene
Nomenclature
Description and references
Opioid analgesic. Prepn of racemate: Pohland, Sullivan, J. Am. Chem. Soc. 75, 4458 (1953); Pohland, US 2728779 (1955 to Lilly). Prepn of (+)-form: Pohland, Sullivan, J. Am. Chem. Soc. 77, 3400 (1955). Stereochemistry: Sullivan et al., J. Org. Chem. 28, 2381 (1963); Casy, Myers, J. Pharm. Pharmacol. 16, 455 (1964). Stereospecific synthesis: Pohland et al., J. Org. Chem. 28, 2483 (1963). Metabolism: S. L. Due et al., Biomed. Mass Spectrom. 3, 217 (1976). The α-dl- and d-diastereoisomers possess marked analgesic activity in contrast to the β-diastereoisomers which are substantially inactive. Toxicity: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971); J. L. Emerson et al., ibid. 19, 445 (1971). Comprehensive description: B. McEwan, Anal. Profiles Drug Subs. 1, 301-318 (1972). Symposium on pharmacology, toxicology, and clinical efficacy of propoxyphene alone and in combination with acetaminophen: Hum. Toxicol. 3, Suppl., 1S-238S (1984).
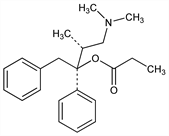
Properties
Crystals from petr ether, mp 75-76°. [α]D25 +67.3° (c = 0.6 in chloroform).Derivative
Hydrochloride.Nomenclature
Properties
Bitter crystals from methanol + ethyl acetate, mp 163-168.5°. [α]D25 +59.8° (c = 0.6 in water). Sol in water, alc, chloroform, acetone. Practically insol in benzene, ether. LD50 in mice, rats (mg/kg): 28, 15 i.v.; 111, 58 i.p.; 211, 134 s.c.; 282, 230 orally (Emerson).Derivative
Napsylate monohydrate.Nomenclature
Properties
Odorless, white crystalline powder; bitter taste. Sol in methanol, ethanol, chloroform, acetone; very slightly sol in water. LD50 orally in female rats: 990 mg/kg (Goldenthal).Derivative
α-l-Form see Levopropoxyphene.Derivative
α-dl-Form.Nomenclature
Racemic propoxyphene; diméprotane.Derivative
α-dl-Form hydrochloride.Properties
Crystals from methanol + ethyl acetate, mp 170-171°. Soluble in water, alcohol, chloroform. Practically insol in benzene, ether.Derivative
β-dl-Form.Properties
Crystals from acetone + ether. mp 187-188°. More soluble than the α-form.Note
Bulk dextropropoxyphene (non-dosage forms) is a controlled substance (opiate): 21 CFR, 1308.12; dextropropoxyphene is a controlled substance (narcotic): 21 CFR, 1308.14.Therapeutic Category
Analgesic.
Keywords
Analgesic; Opioids; Diphenylpropylamine Derivatives