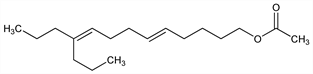
7870. Propylure
Nomenclature
Description and references
Once thought to be the sex pheromone of the pink bollworm moth, Pectinophora gossypiella (Saunders), a destructive cotton pest: H. E. Hummel et al., Science 181, 873 (1973), cf. Gossyplure. Reported as the first known natural product possessing di-n-propyl branching. Isoln, structure, and synthesis: Jones et al., Science 152, 1516 (1966). Improved total syntheses: Pattenden, J. Chem. Soc. C 1968, 2385; Meyers, Collington, Tetrahedron 27, 5979 (1971); Vig et al., J. Indian Chem. Soc. 50, 39 (1973); K. Utimoto et al., Tetrahedron Lett. 1975, 4233. Photochemical synthesis: Kossanyi et al., ibid. 1973, 3459. Stereoselective synthesis: A. Alexakis et al., ibid. 1978, 2027. trans-Propylure is rendered inactive by the presence of >15% of the cis-isomer: Jacobson, Science 163, 190 (1969).