7877. Prostaglandin E2
Nomenclature
Description and references
The most common and most biologically potent of mammalian prostaglandins. Isoln from sheep prostate: S. Bergstr"om, J. Sj"ovall, GB 851827; eidem, US 3598858 (1960, 1971); from sheep seminal vesicle tissue: S. Bergstr"om et al., Acta Chem. Scand. 16, 501 (1962). Total synthesis of the dl-form: W. P. Schneider, Chem. Commun. 1969, 304; E. J. Corey et al., J. Am. Chem. Soc. 91, 5675 (1969); E. J. Corey et al., Tetrahedron Lett. 1970, 307; W. P. Schneider, DE 2011969 (1970 to Upjohn), C.A. 74, 87486n (1971); J. Fried et al., J. Am. Chem. Soc. 94, 4342 (1972). Synthesis of naturally occurring form: E. J. Corey et al., ibid. 92, 397, 2586 (1970); J. B. Heather et al., Tetrahedron Lett. 1973, 2313; from Plexaura homomalla prostaglandin intermediates: G. L. Bundy et al., J. Am. Chem. Soc. 94, 2123 (1972); W. P. Schneider et al., Chem. Commun. 1973, 254. Biosynthesis: D. A. Van Dorp et al., Biochim. Biophys. Acta 90, 204 (1964); S. Bergstr"om et al., ibid. 207; NL 6505799 (1965 to Unilever), C.A. 65, 7584h (1966). Metabolism: E. Anggard, B. Samuelsson, Mem. Soc. Endocrinol., no. 14, 107 (1966); M. Hamberg, B. Samuelsson, J. Biol. Chem. 246, 6713 (1971). Several reviews in Prostaglandin Symp. Worcester Found. Exp. Biol., P. Ramwell, Ed. (Interscience, New York, 1968). For general refs see Prostaglandins.
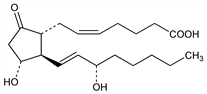
Properties
Natural form, colorless crystals. mp 66-68°. [α]D26 -61° (c = 1 in tetrahydrofuran). Easily dehydrated in soln at pHs <4 or >8.Therapeutic Category
Oxytocic; abortifacient.
Keywords
Abortifacient/Interceptive; Oxytocic; Prostaglandin/Prostaglandin Analog