7878. Prostaglandin F2α
Nomenclature
Description and references
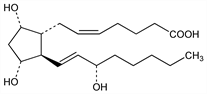
One of the most biologically studied of the primary prostaglandins. Closely related to prostaglandin E2 (PGE2) in that both prostaglandins are biosynthesized from the same precursors and that PGF2α is the synthetic reduction product of PGE2. For refs to synthesis of dl and natural forms see Prostaglandin E2. Prepn of the tromethamine salt: W. Morozowich, DE 2126127; idem, US 3657327 (1971, 1972 both to Upjohn). Alternate synthesis of natural PGF2α: Schneider, Murray, J. Org. Chem. 38, 397 (1973); R. B. Woodward et al., J. Am. Chem. Soc. 95, 6853 (1973); G. Stork et al., ibid. 100, 8272 (1978); K. Kondo et al., Tetrahedron Lett. 1978, 3927; N. R. A. Beeley et al., Tetrahedron 37, Suppl. 9, 411 (1981); R. J. Cave et al., J. Chem. Soc. Perkin Trans. 1 1981, 646. Causes vasocontraction and exhibits luteolytic activity; is most commonly associated with its role in pregnancy: Karim et al., J. Obstet. Gynaecol. Br. Commonw. 78, 172 (1971). Metabolism in female subjects: Granstrom, Samuelsson, J. Biol. Chem. 246, 5254 (1971). Toxicity data: T. Fujita et al., Iyakuhin Kenkyu 9, 261 (1978), C.A. 89, 71399k (1978). For general refs see Prostaglandins.
Properties
Natural form, crystals, mp 25-35°. [α]D25 +23.5° (c = 1 in tetrahydrofuran). Freely sol in methanol, abs ethanol, ethyl acetate, chloroform; slightly sol in water. Stable for two years in light resistant containers at 5-15°. Degrades in one week when exposed to sunlight or in three months at 40°. LD50 in rabbits (mg/kg): 2.5-5.0 i.v.; 2.5-5.0 i.m. (Fujita).Derivative
Tromethamine salt.Nomenclature
Properties
White or off-white cystalline powder, mp 100-101°. Readily sol in water to at least 200 mg/ml.Therapeutic Category
Oxytocic; abortifacient.
Therapeutic Category (Veterinary)
Oxytocic.Keywords
Abortifacient/Interceptive; Oxytocic; Prostaglandin/Prostaglandin Analog