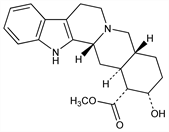
7922. Pseudoyohimbine
Nomenclature
CAS number: 84-37-7
(3β,16α,17α)-17-Hydroxyyohimban-16-carboxylic acid methyl ester. C21H26N2O3; mol wt 354.44.
C 71.16%, H 7.39%, N 7.90%, O 13.54%.
Description and references
Present in the bark of Corynanthe johimbe K. Schum., Rubiaceae. Obtained from the residues of commercial isoln procedures for the manuf of yohimbine: Karrer, Salomon, Helv. Chim. Acta 9, 1059 (1926). Structure and stereochemistry: Janot et al., Bull. Soc. Chim. Fr. 1952, 1085; 1961, 637. Synthesis: van Tamelen et al., J. Am. Chem. Soc. 80, 5006 (1958); 91, 7315 (1969); Stork, Guthikonda, ibid. 94, 5109 (1972).
Properties
Rhombic platelets, mp 293° (corr, Maquenne block), mp 268° (open capillary), browns at 250°. Also reported as crystals from methanol, mp 252-256° (van Tamelen). [α]D19 +27° (pyridine). uv max (methanol): 225, 281, 290 nm (log ε 4.54, 3.86, 3.80).Derivative
Hydrochloride.C21H26N2O3.HCl; mol wt 390.90.
C 64.52%, H 6.96%, N 7.17%, O 12.28%, Cl 9.07%.