7934. Pteropterin
Nomenclature
CAS number: 89-38-3
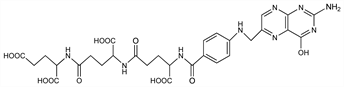
N-[N-[N-[4-[[(2-Amino-1,4-dihydro-4-oxo-6-pteridinyl)methyl]amino]benzoyl]-l-γ-glutamyl]-l-glutamyl]-l-glutamic acid; N-[N-(N-pteroyl-γ-glutamyl)-γ-glutamyl]glutamic
acid; fermentation L. casei Factor; pterolyl-γ-glutamyl-γ-glutamylglutamic acid; pteroyldi-γ-glutamylglutamic acid; pteroyltriglutamic acid; PTGA; Teropterin. C29H33N9O12; mol wt 699.63.
C 49.78%, H 4.75%, N 18.02%, O 27.44%.
Description and references
Isoln from aerobic culture of Corynebacterium: Hutchings et al., J. Am. Chem. Soc. 70, 1 (1948). Identification, structure and synthesis: Boothe et al., ibid. 1099; ibid. 71, 2304 (1949). Prepn: Cosulich, US 2563707 (1951 to Am. Cyanamid).
Properties
Forms a crystalline barium salt.Derivative
Monohydrate.Properties
Crystals from water adjusted to pH 2.8 with HCl and contg some NaCl. Has the general properties of a polypeptide. Absorption spectrum, Hutchings, loc. cit. Soly in water at 5°: 0.10 mg/ml; at 80°: 3.00 mg/ml. Sol in NaOH solns.Derivative
Methyl ester.Properties
Crystals from methanol contg NaCl. Soly in water at 5°: 0.12 mg/ml; at 80°: 1.00 mg/ml; in methanol at -5°: 0.30 mg/ml; at 60°: 5.00 mg/ml.Therapeutic Category
Formerly as antineoplastic.
Keywords
Antineoplastic; Antimetabolites; Folic Acid Analogs/Antagonists