7945. Purpurin
Nomenclature
CAS number: 81-54-9
1,2,4-Trihydroxy-9,10-anthracenedione; 1,2,4-trihydroxyanthraquinone; C.I. Natural Red 8; C.I. Natural Red 16; C.I. 58205; C.I. 75410. C14H8O5; mol wt 256.21.
C 65.63%, H 3.15%, O 31.22%.
Description and references
Occurs as glycoside in the madder root (Rubia tinctorum L., Rubiaceae) of commerce. Is formed during storage; no appreciable amount in the fresh root: Hill, Richter, J. Chem. Soc. 1936, 1714. Although a dye itself, it is usually considered as an undesirable contamimant of alizarin extracted from madder. May be prepd from alizarin by oxidation with ammonium persulfate: Wacker, J. Prakt. Chem. [2] 54, 90 (1896); also by Friedel-Crafts condensation of hydroxyhydroquinone with phthalic anhydride: Dimroth, Fick, Ann. 411, 321 (1916).
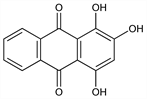
Properties
Long orange needles with 1 H2O from dil alcohol, anhydr at 100°. Anhydr red needles from abs alcohol or by sublimation around 150° in high vacuum (less than 2 mm Hg). mp 257°. Absorption spectrum: Meek, J. Chem. Soc. 111, 969 (1917); Ezaby, J. Chem. Soc. B 1970, 1293. More sol in boiling water than alizarin (yellow color with yellowish hue). Freely sol in alcohol (red), in ether (intensely yellow with fluorescence). Soluble in benzene, toluene, xylene (dark yellow), in boiling alum soln (red).Derivative
2-Methyl ether.C15H10O5; mol wt 270.24.
C 66.67%, H 3.73%, O 29.60%.
Properties
Orange crystals from benzene, mp 240°.Derivative
2,4-Dimethyl ether.C16H12O5; mol wt 284.26.
C 67.60%, H 4.26%, O 28.14%.