7946. Purpurogallin
Nomenclature
CAS number: 569-77-7
2,3,4,6-Tetrahydroxy-5H-benzocyclohepten-5-one. C11H8O5; mol wt 220.18.
C 60.00%, H 3.66%, O 36.33%.
Description and references
The aglycone of several glycosides from various nutgalls. Prepn by oxidation of pyrogallol: Perkin, Steven, J. Chem. Soc. 83, 192 (1903); Perkin, ibid. 101, 803 (1912); Nierenstein, Spiers, Ber. 46, 3151 (1913); Evans, Dehn, J. Am. Chem. Soc. 52, 3647 (1930). Structure: Willst"atter, Heiss, Ann. 433, 17 (1923); Barltrop, Nicholson, J. Chem. Soc. 1948, 116. Synthesis: Caunt et al., ibid. 1950, 1631; 1951, 1313.
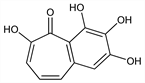
Properties
Deep red needles from glacial acetic acid, dec 274-275°. Sparingly sol in most solvents.Derivative
Diglucoside.Nomenclature
Dryophantin.C23H28O15; mol wt 544.46.
C 50.74%, H 5.18%, O 44.08%.